
Amixture of noble gases [helium (mw 4), argon (mw 40), krypton (mw 83.8), and xenon (mw 131.3)] is at a total pressure of 150 kpa, and a temperature of 500 k. the mixture has the following composition in mole fraction: 0.25 helium, 0.25 argon, 0.25 krypton. determine: (a) the mass fraction of helium. (b) the average molecular weight of the mixture. (c) the total molar concentration. (d) the mass density.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 07:30
Label a-f based on the table using c for concentrated and d for dilute
Answers: 2

Chemistry, 22.06.2019 09:20
What happened to the amount of carbon dioxide in the atmosphere from 2010–2017?
Answers: 1

Chemistry, 22.06.2019 21:30
Under which circumstances are kp and kc equal for the reaction aa(g)+bb(g)⇌cc(g)+dd(g)?
Answers: 2

You know the right answer?
Amixture of noble gases [helium (mw 4), argon (mw 40), krypton (mw 83.8), and xenon (mw 131.3)] is a...
Questions


Social Studies, 29.06.2019 10:00




History, 29.06.2019 10:00







Social Studies, 29.06.2019 10:00

Mathematics, 29.06.2019 10:00


Health, 29.06.2019 10:00

History, 29.06.2019 10:00

History, 29.06.2019 10:00

Health, 29.06.2019 10:00

 = 1 g of HeliumArgon: 0,25 moles ×
= 1 g of HeliumArgon: 0,25 moles ×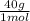 = 10 g of ArgonKrypton: 0,25 moles ×
= 10 g of ArgonKrypton: 0,25 moles ×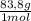 = 20,95 g of krypton Xenon: 0,25 moles ×
= 20,95 g of krypton Xenon: 0,25 moles ×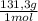 = 32,825 g of Xenon
= 32,825 g of Xenon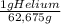 × 100 = 1,6%
× 100 = 1,6%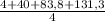 = 64,775 g/mol
= 64,775 g/mol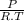 = M
= M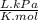
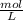 ×
× 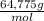 ×
× 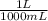 =
=


