
Chemistry, 16.09.2019 22:00 ericahale3971
Suppose 6.54g of potassium bromide is dissolved in 50.ml of a 0.70 m aqueous solution of silver nitrate. calculate the final molarity of bromide anion in the solution. you can assume the volume of the solution doesn't change when the potassium bromide is dissolved in it.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 00:40
Which is a difference between molecular compounds and ionic compounds? select the correct answer below: question 5 options: molecular compounds typically form between a metal and a nonmetal, while ionic compounds typically form between nonmetals. molecular compounds result from the transfer of electrons between atoms to form ions, while ionic compounds result from the sharing of electrons between neutral atoms. molecular compounds are formed of discrete, neutral molecules, while ionic compounds are formed of large repeating arrays of opposite charges. molecular compounds have high melting points and high boiling points, while ionic
Answers: 3

Chemistry, 22.06.2019 13:30
Which of the following natural processes is most likely to support the formation of an underwater sinkhole? a pollution buildup from deposited minerals b limestone cave collapsing due to changes in sea level c erosion of large amounts of sand moved by ocean waves d oxidation of rock formed by chemical weathering
Answers: 1

Chemistry, 22.06.2019 21:30
If 22.5 of nitrogen at 748 mm hg are compressed to 725 mm hg at constant temperature. what is the new volume?
Answers: 1

Chemistry, 23.06.2019 00:50
The chemical formula for emerald is be3al2(sio3)6.an emerald can be decided as
Answers: 3
You know the right answer?
Suppose 6.54g of potassium bromide is dissolved in 50.ml of a 0.70 m aqueous solution of silver nitr...
Questions

Biology, 04.12.2020 21:10

Mathematics, 04.12.2020 21:10


Chemistry, 04.12.2020 21:10

Social Studies, 04.12.2020 21:10

Mathematics, 04.12.2020 21:10

History, 04.12.2020 21:10


Mathematics, 04.12.2020 21:10

Computers and Technology, 04.12.2020 21:10


Mathematics, 04.12.2020 21:10

English, 04.12.2020 21:10


English, 04.12.2020 21:10

Mathematics, 04.12.2020 21:10

Mathematics, 04.12.2020 21:10


Mathematics, 04.12.2020 21:10

SAT, 04.12.2020 21:10

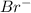 ]=0.4M
]=0.4M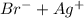 ⇄
⇄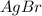 Kps for this reaction is
Kps for this reaction is 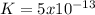 , this constant indicates if the reaction can happen spontaneously or not. in this case, a small value of Kps means that precipitate can be formed.
, this constant indicates if the reaction can happen spontaneously or not. in this case, a small value of Kps means that precipitate can be formed.![[Br^{-}]=6.54gKBr.\frac{1molKBr}{119gKBr}.\frac{1molBr^{-} }{1molKBr} .\frac{1}{0.05L} =1.10M](/tpl/images/0234/3240/bfec1.png)
![Qps=[Br^{-}][Ag^{+}]](/tpl/images/0234/3240/78356.png) =
=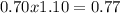
![[Br^{-}]=\frac{(\frac{1.1mol}{L}.0.05L-\frac{0.70mol}{L}.0.05L}{0.05L} =0.4M](/tpl/images/0234/3240/ff056.png)


