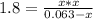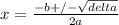Chemistry, 16.09.2019 19:30 gracebuffum
Phosphorus pentachloride decomposes according to the chemical equation pcl5(g)↽−−⇀pcl3(g)+cl2(g)kc=1.80 at 250 ∘c a 0.157 mol sample of pcl5(g) is injected into an empty 2.50 l reaction vessel held at 250 ∘c. calculate the concentrations of pcl5(g) and pcl3(g) at equilibrium.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 00:30
What is the most stable monatomic ion formed from nitrogen
Answers: 2

Chemistry, 22.06.2019 01:30
What is the value of keq for the reaction expressed in scientific notation
Answers: 1

Chemistry, 22.06.2019 10:30
What woukd most likely be the transmittance at a 0.70 m solution of solute a? a) 7.6%b) 1.1%c)4.0%d)4.6%
Answers: 1

Chemistry, 22.06.2019 10:30
Use this information to determine the number of calends electrons in the atoms. which of the following correctly compares the stability of the two atoms? a) both are unreactive b) both are highly reactive c) a is unreactive and d is reactive d) a is reactive and d is unreactive
Answers: 2
You know the right answer?
Phosphorus pentachloride decomposes according to the chemical equation pcl5(g)↽−−⇀pcl3(g)+cl2(g)kc=1...
Questions

History, 21.04.2020 23:36






Chemistry, 21.04.2020 23:36

Mathematics, 21.04.2020 23:36


Social Studies, 21.04.2020 23:36

Mathematics, 21.04.2020 23:36



Mathematics, 21.04.2020 23:37



Mathematics, 21.04.2020 23:37



![Kc = \frac{[C]^cx[D]^d}{[A]^ax[B]^b}](/tpl/images/0233/9746/d3f86.png)
![Kc = \frac{[PCl3]x[Cl2]}{[PCl5]}](/tpl/images/0233/9746/20f28.png)