
Phosphorus forms several compounds with oxygen, including diphosphorus trioxide and diphosphorus pentoxide. a decomposition of a sample of diphosphorus trioxide forms 1.29 g phosphorus to every 1.00 g oxygen. the decomposition of a sample of diphosphorus pentoxide forms 0.775 g phosphorus to every 1.00 g oxygen. show that these results are consistent with the law of multiple proportions.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 02:30
The is a particle with one unit of positive charge a. proton b. positron c. electron d. nucleus awnser quick it is a important science test!
Answers: 2


Chemistry, 22.06.2019 14:00
How does the presence of oxygen affect the chemical pathways used to extract energy from glucose?
Answers: 3

Chemistry, 22.06.2019 14:30
What type(s) of intermolecular forces are expected between ch3ch2cooh molecules? dipole forces, induced dipole forces, hydrogen bonding
Answers: 1
You know the right answer?
Phosphorus forms several compounds with oxygen, including diphosphorus trioxide and diphosphorus pen...
Questions

Health, 25.06.2019 12:00

Mathematics, 25.06.2019 12:00

History, 25.06.2019 12:00

English, 25.06.2019 12:00



Mathematics, 25.06.2019 12:00


History, 25.06.2019 12:00

Computers and Technology, 25.06.2019 12:00

Chemistry, 25.06.2019 12:00


Mathematics, 25.06.2019 12:00


Mathematics, 25.06.2019 12:00

Mathematics, 25.06.2019 12:00




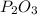 the ratio between P and O is
the ratio between P and O is 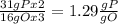 , that is consistent with the experimental result.
, that is consistent with the experimental result.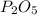 the ratio between P and O is
the ratio between P and O is 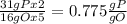 that is also consistent with experimental results. In conclusion we can say that the results obtained are consistent with the law of multiple proportions.
that is also consistent with experimental results. In conclusion we can say that the results obtained are consistent with the law of multiple proportions.

