
Chemistry, 16.09.2019 17:10 briweaver9993
Titanium dioxide, tio₂, reacts with carbon and chlorine to give gaseous ticl₄: tio₂+2c+2ci₂−tici₄+2co the reaction of 7.39 kg titanium dioxide with excess c and cl₂ gives 14.24 kg titanium tetrachloride. calculate the theoretical yield of ticl₄ (assuming complete reaction) and its percentage yield.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 20:30
The speed of light is around 6.706×10^8 miles per hour. what is the speed of light in units of miles per minute?
Answers: 2

Chemistry, 21.06.2019 22:30
How much heat is released upon converting one mole of steam (18.0 g) from 100.0 ∘c to water at 25.0 ∘c? show work and constants, trying to figure out how it works. only given the heat capacity for steam and water so try to only use that
Answers: 1

Chemistry, 22.06.2019 10:30
When the speed of the bottle is 2 m/s, the average maximum height of the beanbag is m.
Answers: 2

Chemistry, 22.06.2019 15:50
How many moles of potassium hydroxide are needed to completely react with 2.94 moles of aluminum sulfate
Answers: 1
You know the right answer?
Titanium dioxide, tio₂, reacts with carbon and chlorine to give gaseous ticl₄: tio₂+2c+2ci₂−tici₄+2...
Questions

Social Studies, 17.01.2021 02:00


Arts, 17.01.2021 02:00



Health, 17.01.2021 02:00



Mathematics, 17.01.2021 02:00

Arts, 17.01.2021 02:00


Mathematics, 17.01.2021 02:00





Mathematics, 17.01.2021 02:00


Mathematics, 17.01.2021 02:00

Mathematics, 17.01.2021 02:00

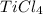 and its percentage yield is 81.0%
and its percentage yield is 81.0%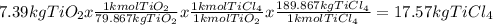
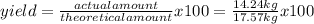 =81.0%
=81.0%

