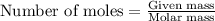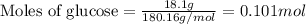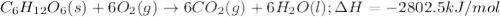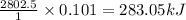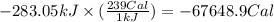Chemistry, 14.09.2019 10:20 GalaxyCraft4991
the oxidation of the sugar glucose, c6h12o6, is described by the following equation. c6h12o6(s) + 6 o2(g) → 6 co2(g) + 6 h2o(l) δh = −2802.5 kj/mol the metabolism of glucose gives the same products, although the glucose reacts with oxygen in a series of steps in the body.
(a) how much heat in kilojoules can be produced by the metabolism of 18.1 g of glucose?
(b) how many calories can be produced by the metabolism of 18.1 g of glucose?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 06:00
One of the few xenon compounds that form is cesium xenon heptafluoride (csxef7). how many moles of csxef7 can be produced from the reaction of 13.0 mol cesium fluoride with 12.5 mol xenon hexafluoride? csf(s) + xef6(s) csxef7(s)
Answers: 1


Chemistry, 22.06.2019 07:50
In which situation can a mixture always be called a solution
Answers: 3

Chemistry, 22.06.2019 08:30
What method(s) do plants use to obtain nitrogen? select all that apply. absorb it from the atmosphere use bacteria to convert nitrogen to usable form obtain usable nitrogen compounds from the soil absorb nitrogen from water taken in at the roots
Answers: 3
You know the right answer?
the oxidation of the sugar glucose, c6h12o6, is described by the following equation. c6h12o6(s) + 6...
Questions

Mathematics, 27.08.2019 10:00

Mathematics, 27.08.2019 10:00


Biology, 27.08.2019 10:00




Mathematics, 27.08.2019 10:00

Chemistry, 27.08.2019 10:00

Computers and Technology, 27.08.2019 10:00


Mathematics, 27.08.2019 10:00

Mathematics, 27.08.2019 10:00

Biology, 27.08.2019 10:00

Biology, 27.08.2019 10:00

Chemistry, 27.08.2019 10:00

Physics, 27.08.2019 10:00

Biology, 27.08.2019 10:00

History, 27.08.2019 10:00

Biology, 27.08.2019 10:00