
Chemistry, 14.09.2019 10:10 mwms2018sg
You have a 0.5 l of a 1.0 m buffer solution with pka = 5.14 and current ph = 5.23. calculate the new ph when 1.50 ml of 5.00 m hcl is added. be mindful of units. (how can you tell if your answer makes sense? ) ph = pka + 109 u base 5.23 - 5.14+ log ( )

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 16:50
Answer asap need it by wednesday morning calculate the ph of 0.02m hcl best answer will be brainliest
Answers: 1

Chemistry, 22.06.2019 21:30
Harry lives in a city, and he has a lung condition known as asthma. on certain days, harry has to stay inside because pollutants in the air make it difficult for him to breathe. which of these pollution sources are nonpoint sources that might bother harry if he goes outside? choose the two that apply.
Answers: 3

Chemistry, 23.06.2019 00:30
If there are 3.5 moles of koh, how many moles of naoh can be produced? question 1 options: a)3.0 moles naoh b)3.5 moles naoh c)1 moles naoh d)9 moles naoh
Answers: 1

Chemistry, 23.06.2019 03:30
Calculate the ph of a .10m nh4cl solution. the kb value for nh3 is 1.8×10^-5
Answers: 1
You know the right answer?
You have a 0.5 l of a 1.0 m buffer solution with pka = 5.14 and current ph = 5.23. calculate the new...
Questions

Mathematics, 21.06.2021 18:00

Mathematics, 21.06.2021 18:00

English, 21.06.2021 18:00


History, 21.06.2021 18:00


Chemistry, 21.06.2021 18:00


Mathematics, 21.06.2021 18:00

English, 21.06.2021 18:00

Physics, 21.06.2021 18:00


Social Studies, 21.06.2021 18:00

English, 21.06.2021 18:00


Mathematics, 21.06.2021 18:00

Mathematics, 21.06.2021 18:00

Mathematics, 21.06.2021 18:00

English, 21.06.2021 18:00

Mathematics, 21.06.2021 18:00

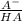
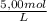 ÷ 0,5 L = 0,015 M of HCl
÷ 0,5 L = 0,015 M of HCl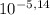
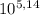 =
= ![\frac{[0,015M -x][0,55M-x]}{[0,45M-x]}](/tpl/images/0231/2479/0fe82.png)


