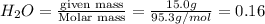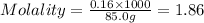Chemistry, 14.09.2019 08:30 trillralyn4060
Calculate the molality of a 150% by mass solution of mgcl, fw-95.3 g/mol in h. o. the density of tis solution is 1.127 gim 0.0134 m 0.157 m 1.58 m 1.86 m 11.8 m igator delete backspace u 10 pilli

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 02:00
What is the maximum number of electrons that an atomic orbital can contain?
Answers: 1

Chemistry, 22.06.2019 06:00
Which of the following did jj thompson discover about atoms? a)an atom has an internal structure. b) atoms are tiny indivisible particles. c)electrons orbit the nucleus of an atom. d) the nucleus of an atom contains protons and neutrons.
Answers: 2

Chemistry, 22.06.2019 06:30
Three cards with holes are arranged in a straight line. a light is shined through the first card’s hole and travels through all three cards. what does this tell you about light rays? a) that light is reflected b) that light is refractive c) that light travels in a straight line d) that light does not travel in a straight line
Answers: 1

Chemistry, 22.06.2019 10:10
For the reaction, 4 a(g) + 3 b(g) => 2 c(g), the following data were obtained at constant temperature. experiment initial[a],mol/l initial [b],mol/l initial rate,m/min 1 0.200 0.150 5.00 2 0.400 0.150 10.0 3 0.200 0.300 10.0 4 0.400 0.300 20.0 which of the following is the correct rate law for the reaction? 1. rate = k[a]2[b]2 2. rate = k[a][b] 3. rate = k[a]2[b] 4. rate = k[a][b]2
Answers: 3
You know the right answer?
Calculate the molality of a 150% by mass solution of mgcl, fw-95.3 g/mol in h. o. the density of tis...
Questions


Biology, 01.08.2019 01:30


Mathematics, 01.08.2019 01:30


Spanish, 01.08.2019 01:30

English, 01.08.2019 01:30


Mathematics, 01.08.2019 01:30









Computers and Technology, 01.08.2019 01:30


Mathematics, 01.08.2019 01:30

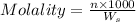
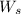 = weight of solvent in g
= weight of solvent in g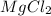 is present in 100 g of solution
is present in 100 g of solution