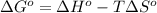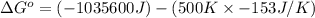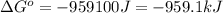Use the given data at 500 k to calculate δg°for the reaction
2h2s(g) + 3o2(g) → 2h2o(g) + 2so2...

Chemistry, 14.09.2019 08:20 87haymaker
Use the given data at 500 k to calculate δg°for the reaction
2h2s(g) + 3o2(g) → 2h2o(g) + 2so2(g)
substance h2s(g) o2(g) h2o(g) so2(g)
δh°f(kj/mol) -21 0 -242 -296.8
s°(j/k·mol) 206 205 189 248

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 04:40
*will mark you brainliest + 15 points ** why does the equilibrium of a system shift when the pressure is increased? a. to maximize the stress on the system b. to stop restoring equilibrium to the system c. to increase the total moles of gas in the system d. to decrease the total moles of gas in the system
Answers: 3


Chemistry, 22.06.2019 11:00
The diagram below shows the different phase transitions that occur in matter. which arrow represents the transition in which dew is formed?
Answers: 1

Chemistry, 22.06.2019 13:00
One of the hopes for solving the world's energy problem is to make use of the fusion reaction 21h +31h --> 42he + 10n + energy how much energy is released when 1 mol of deuterium is fused with 1 mol of tritium according to the above reaction? the masses of the atoms and the neutrons are as follows: 21h = 2.0140 amu 31h = 3.01605 amu 42he = 4.002603 amu 10n = 1.008665 amu. the speed of light is 2.9979 x 108 m/s.
Answers: 1
You know the right answer?
Questions


Physics, 04.12.2020 01:20

History, 04.12.2020 01:20

Chemistry, 04.12.2020 01:20

History, 04.12.2020 01:20

Social Studies, 04.12.2020 01:20



Physics, 04.12.2020 01:20

Social Studies, 04.12.2020 01:20


Mathematics, 04.12.2020 01:20

Mathematics, 04.12.2020 01:20


English, 04.12.2020 01:20


Mathematics, 04.12.2020 01:20

Mathematics, 04.12.2020 01:20


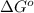 for the reaction is -959.1 kJ
for the reaction is -959.1 kJ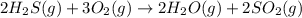
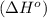 .
.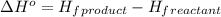
![\Delta H^o=[n_{H_2O}\times \Delta H_f^0_{(H_2O)}+n_{SO_2}\times \Delta H_f^0_{(SO_2)}]-[n_{H_2S}\times \Delta H_f^0_{(H_2S)}+n_{O_2}\times \Delta H_f^0_{(O_2)}]](/tpl/images/0231/1095/c2362.png)
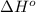 = enthalpy of reaction = ?
= enthalpy of reaction = ?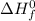 = standard enthalpy of formation
= standard enthalpy of formation![\Delta H^o=[2mole\times (-242kJ/mol)+2mole\times (-296.8kJ/mol)}]-[2mole\times (-21kJ/mol)+3mole\times (0kJ/mol)]](/tpl/images/0231/1095/0563e.png)
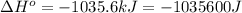
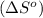 .
.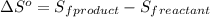
![\Delta S^o=[n_{H_2O}\times \Delta S_f^0_{(H_2O)}+n_{SO_2}\times \Delta S_f^0_{(SO_2)}]-[n_{H_2S}\times \Delta S_f^0_{(H_2S)}+n_{O_2}\times \Delta S_f^0_{(O_2)}]](/tpl/images/0231/1095/f7046.png)
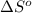 = entropy of reaction = ?
= entropy of reaction = ?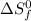 = standard entropy of formation
= standard entropy of formation![\Delta S^o=[2mole\times (189J/K.mol)+2mole\times (248J/K.mol)}]-[2mole\times (206J/K.mol)+3mole\times (205J/K.mol)]](/tpl/images/0231/1095/0797b.png)
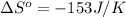
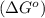 .
.