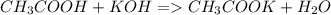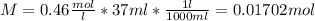25g of vinegar (a solution containing acetic acid) was addedto
a flask containing an indicator...

Chemistry, 13.09.2019 23:10 asialovepink2321
25g of vinegar (a solution containing acetic acid) was addedto
a flask containing an indicator. 37ml of .46m koh solution wasadded
to the system from a burette to reach the equivalence point. what is
the percentage by mass of vinegar that is aceticacid?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 07:10
Remember to use the proper number of significant figures and leading zeros in all calculations.gelatin has a density of 1.27 g/cm³. if you have a blob of gelatin dessert that fills a 2.0 liter bottle, what is its mass? 2540 g2500 g3.9 x 10-43.937x 10-4
Answers: 3

Chemistry, 22.06.2019 11:50
The chemical bond connecting one nucleotide with the next one along the nucleic acid chain is called a
Answers: 3

Chemistry, 22.06.2019 13:30
Which statements are true concerning mineral formation? check all that apply. the slower the cooling, the larger the crystals. the faster the cooling, the smaller the crystals. crystals formed from magma are smaller than crystals formed from lava. minerals can only form in solutions when the solution is heated deep underground. when a solution cools, elements and compounds leave the solution and crystallize as minerals. minerals formed from hot water solutions can form narrow channels in the surrounding rock.
Answers: 1

You know the right answer?
Questions

Mathematics, 28.10.2020 02:00

Mathematics, 28.10.2020 02:00


Mathematics, 28.10.2020 02:00

Chemistry, 28.10.2020 02:00




Mathematics, 28.10.2020 02:00

Mathematics, 28.10.2020 02:00


Mathematics, 28.10.2020 02:00


History, 28.10.2020 02:00

Computers and Technology, 28.10.2020 02:00

Mathematics, 28.10.2020 02:00


Mathematics, 28.10.2020 02:00

English, 28.10.2020 02:00

Mathematics, 28.10.2020 02:00