

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 14:50
Consider the following multistep reaction: a b→ab(slow) a ab→a2b(fast)−−−−−−−−−−−−−−−−− 2a b→a2b(overall) based on this mechanism, determine the rate law for the overall reaction. express your answer in standard masteringchemistry format. for example, if the rate law is k[a]3[b]2 type k*[a]^3*[b]^2
Answers: 3

Chemistry, 22.06.2019 15:30
How does a large body of water, such as the ocean, influence climate?
Answers: 1

Chemistry, 22.06.2019 21:50
Given the data below for the reaction, 2 a + 2 b + 4 c => d + e + 3 f, the reaction is fill in the [ ] order in a, fill in the [ ] order in b, fill in the [ ] order in c and fill in the [ ] order overall. (use the words "first, second, third, fourth" to fill each blank)experimentinitial conc of a, mol/l initial conc of b, mol/l initial conc of c, mol/l initial rate, mol/l.s1 0.1 0.1 0.2 2 x 10-32 0.2 0.3 0.2 6 x 10-33 0.3 0.1 0.2 2 x 10-34 0.4 0.3 0.4 1.2 x 10-2
Answers: 2

Chemistry, 22.06.2019 23:00
In the reaction h2co3 (aq) + 3nh3 (aq) = 2 nh4+ (aq) + co3 2-, how many electrons are transferred?
Answers: 3
You know the right answer?
A1004.0g sample of calcium carbonate that is 95.0% pure
gives225l of co2 at stp when reacted w...
gives225l of co2 at stp when reacted w...
Questions

Computers and Technology, 24.09.2020 08:01

Biology, 24.09.2020 08:01






Mathematics, 24.09.2020 08:01

Mathematics, 24.09.2020 08:01

Mathematics, 24.09.2020 08:01

English, 24.09.2020 08:01

History, 24.09.2020 08:01



Mathematics, 24.09.2020 08:01



Mathematics, 24.09.2020 08:01

Mathematics, 24.09.2020 08:01

Biology, 24.09.2020 08:01

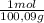 =
= 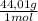 = 419,4 g of CO₂
= 419,4 g of CO₂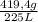 = 1,86 g/L
= 1,86 g/L

