
Chemistry, 13.09.2019 22:30 sosick90501
Question: dimethylhydrazine, the
fuel used in the apollo lunar descentmodule, has a molar mass of
60.10 g/mol. it is made up of carbon, hydrogen, and nitrogen atoms.
the combustion of 2.859g of the fuelin excess oxygen yields 4.190g
of carbon dioxideand 3.428g ofwater. what are the simplest and
molecular formulas fordimethylhydrazine?

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 18:40
Determine the mass of fuel required for the expected energy consumption in the united states for the next ten years. energy use per person per year in the united states = 3.5 x 1011joules base calculations on current population of 310,000,000.
Answers: 2

Chemistry, 21.06.2019 21:30
Which substances have the lowest melting points: ionic covalent, or metallic
Answers: 1

Chemistry, 22.06.2019 17:00
How can a give a full method for the experiment of separating sand from water by filtration? 1-materials 2-steps 3-conclusion also for water and salt separated by the evaporation or distillation process
Answers: 1

Chemistry, 22.06.2019 18:10
Consider the following reaction at equilibrium: c(s)+h2o(g)⇌co(g)+h2(g) predict whether the reaction will shift left, shift right, or remain unchanged upon each of the following disturbances. a) c is added to the reaction mixture. b) h2ois condensed and removed from the reaction mixture c) co is added to the reaction mixture d) h2 is removed from the reaction mixture.
Answers: 3
You know the right answer?
Question: dimethylhydrazine, the
fuel used in the apollo lunar descentmodule, has a molar mass...
fuel used in the apollo lunar descentmodule, has a molar mass...
Questions








Business, 02.07.2019 07:00


Biology, 02.07.2019 07:00




Mathematics, 02.07.2019 07:00

Mathematics, 02.07.2019 07:00

Mathematics, 02.07.2019 07:00



Mathematics, 02.07.2019 07:00

Health, 02.07.2019 07:00

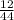 x 4.19g = 1.14g
x 4.19g = 1.14g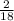 x 3.428g = 0.38g
x 3.428g = 0.38g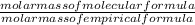 =
= 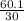 = 2
= 2

