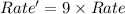Consider the following reaction: 2 no(g) + 2h2(g) → n2(g) + 2 h2o(g) the rate law for this reaction is first order in h2 and second order in no. what would happen to the rate if the initial concentration of no tripled while all other factors stayed the same? the rate will increase by a factor of 9. the rate will decrease by a factor of 3. the rate will double. the rate will triple. the rate will remain constant.

Answers: 3
Another question on Chemistry




Chemistry, 22.06.2019 22:30
Rank the four gases (air, exhaled air, gas produced from from decomposition of h2o2, gas from decomposition of nahco3) in order of increasing concentration of co2
Answers: 1
You know the right answer?
Consider the following reaction: 2 no(g) + 2h2(g) → n2(g) + 2 h2o(g) the rate law for this reaction...
Questions


History, 29.06.2019 19:00



Spanish, 29.06.2019 19:00



History, 29.06.2019 19:00

Mathematics, 29.06.2019 19:00

Geography, 29.06.2019 19:00



English, 29.06.2019 19:00

History, 29.06.2019 19:00

Chemistry, 29.06.2019 19:00

Biology, 29.06.2019 19:00

Mathematics, 29.06.2019 19:00

History, 29.06.2019 19:00

Mathematics, 29.06.2019 19:00

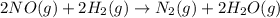
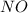 = 2
= 2 = 1
= 1![Rate=k[NO]^2[H_2]^1](/tpl/images/0229/1480/39530.png)
![Rate'=k[3\times NO]^2[H_2]^1](/tpl/images/0229/1480/e80a2.png)
![Rate'=k[3]^2[NO]^2[H_2]^1](/tpl/images/0229/1480/b88df.png)
![Rate'=k\times 9[NO]^2[H_2]^1](/tpl/images/0229/1480/d2dcb.png)