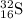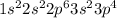Chemistry, 11.09.2019 21:30 nxusasmangaliso7596
Refer to the following elements to answer the questions below. 126c168o11h147n3216s3115p, where the upper index is atomic mass and the lower index is atomic number. how many electrons will a single atom of sulfur with no charge and no bonds have in its valence shell?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 09:00
An excess of lithium oxide undergoes a synthesis reaction with water to produce lithium hydroxide li2o+h2o→2lioh if 1.05 g of water reacted, what is the theoretical yield of lithium hydroxide? a) 5.83 x 10–2 g lioh b) 1.17 x 10–1 g lioh c) 2.79 x 100 g lioh d) 1.40 x 100 g lioh
Answers: 1

Chemistry, 22.06.2019 14:30
For the reaction shown, find the limiting reactant for each of the following initial amounts of reactants. 4al(s)+3o2(g)→2al2o3(s) a) 1 molal, 1 mol o2 b) 4 molal, 2.6 mol o2 c) 16 molal, 13 mol o2 d) 7.4 molal, 6.5 mol o2
Answers: 3

Chemistry, 22.06.2019 20:00
There are two steps in the usual industrial preparation of acrylic acid, the immediate precursor of several useful plastics. in the first step, calcium carbide and water react to form acetylene and calcium hydroxide: cac2 (s) + 2h2o (g) → c2h2 (g) + caoh2 (s) =δh−414.kj in the second step, acetylene, carbon dioxide and water react to form acrylic acid: 6c2h2 (g) + 3co2 (g) + 4h2o (g) → 5ch2chco2h (g) =δh132.kj calculate the net change in enthalpy for the formation of one mole of acrylic acid from calcium carbide, water and carbon dioxide from these reactions. round your answer to the nearest kj .
Answers: 3

Chemistry, 23.06.2019 04:00
Silver reacts with oxygen to produce silver oxide. (write balanced chemical equation and identify type of chemical reaction.)
Answers: 1
You know the right answer?
Refer to the following elements to answer the questions below. 126c168o11h147n3216s3115p, where the...
Questions


History, 24.12.2020 01:20

Mathematics, 24.12.2020 01:20


Mathematics, 24.12.2020 01:20




Mathematics, 24.12.2020 01:20

Mathematics, 24.12.2020 01:20


Spanish, 24.12.2020 01:20



Spanish, 24.12.2020 01:20

Health, 24.12.2020 01:20


Physics, 24.12.2020 01:20

Chemistry, 24.12.2020 01:20

Mathematics, 24.12.2020 01:20