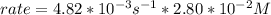Chemistry, 11.09.2019 00:30 nicholasryanencarnac
In carbon tetrachloride proceeds as follows: 2n2o5→4no2+o2. the rate law is first order in n2o5. at 64 ∘c the rate constant is 4.82 ×10−3s−1. part a part complete write the rate law for the reaction. write the rate law for the reaction. rate=2.41×10−3s−1[n2o5] rate=4.82×10−3s−1[n2o5] rate=9.64×10−3s−1[n2o5] rate=4.82×10−3s−1[n2o5]2 previous answers correct part b what is the rate of reaction when [n2o5]= 2.80×10−2 m ?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 09:30
Based on its chemical properties, identify the position of each chemical family on the periodic table.
Answers: 3


Chemistry, 22.06.2019 17:30
Take a look at this dandelion. the yellow flower on the right is pollinated and the seeds on the left are transported by
Answers: 2

Chemistry, 23.06.2019 04:20
Calculate the mass of 0.750 mol of the following substance. na3po4.
Answers: 1
You know the right answer?
In carbon tetrachloride proceeds as follows: 2n2o5→4no2+o2. the rate law is first order in n2o5. at...
Questions



Physics, 20.09.2020 02:01

Mathematics, 20.09.2020 02:01



History, 20.09.2020 02:01

History, 20.09.2020 02:01


Physics, 20.09.2020 02:01

Mathematics, 20.09.2020 02:01

Social Studies, 20.09.2020 02:01

English, 20.09.2020 02:01

Mathematics, 20.09.2020 02:01



Geography, 20.09.2020 02:01



English, 20.09.2020 02:01

![rate= 4.82*10^{-3}s^{-1} * [N2O5]](/tpl/images/0227/3013/f16e4.png)
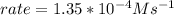
![rate= k * [N2O5]^{x}](/tpl/images/0227/3013/f017a.png)