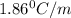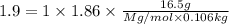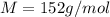Chemistry, 10.09.2019 23:10 blakestuhan
An aqueous solution containing 16.5 g of an unknown molecular (nonelectrolyte) compound in 106.0 g of water was found to have a freezing point of -1.9 ∘c. calculate the molar mass of the unknown compound.

Answers: 3
Another question on Chemistry



Chemistry, 22.06.2019 14:00
Will mark brainliest how many electrons can be held in the energy level n = 4?
Answers: 1

You know the right answer?
An aqueous solution containing 16.5 g of an unknown molecular (nonelectrolyte) compound in 106.0 g o...
Questions




Mathematics, 21.08.2019 11:10



Social Studies, 21.08.2019 11:10

English, 21.08.2019 11:10

Mathematics, 21.08.2019 11:10

English, 21.08.2019 11:10

Computers and Technology, 21.08.2019 11:10

Chemistry, 21.08.2019 11:10

Mathematics, 21.08.2019 11:10

Mathematics, 21.08.2019 11:10




Mathematics, 21.08.2019 11:10

History, 21.08.2019 11:10

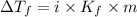
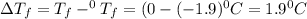 = Depression in freezing point
= Depression in freezing point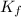 = freezing point constant =
= freezing point constant =