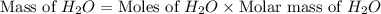Chemistry, 09.09.2019 19:30 Frenchfries13
Consider the following unbalanced chemical equation. c5h12(l) + o2(g) → co2(g) + h2o(l) if 21.9 grams of pentane (c5h12) are burned in excess oxygen, how many grams of h2o will be produced?

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 14:00
Which of the following statements is true? question 4 options: nuclear decay rates vary with the conditions of the reaction, but chemical reaction rates do not. chemical reaction rates vary with the conditions of the reaction, but nuclear decay rates do not. neither chemical reaction rates nor nuclear decay rates vary with the conditions of the reaction. both chemical reaction rates and nuclear decay rates vary with the conditions of the reaction.
Answers: 1

Chemistry, 21.06.2019 15:30
Is this a scientific model? use complete sentences to explain why or why not. a graphic organizer showing the water cycle
Answers: 3

Chemistry, 21.06.2019 22:30
Ibeg i need 20. a reaction produces 4.93 l of oxygen, but was supposed to produce 1 mol of oxygen. what is the percent yield?
Answers: 1

You know the right answer?
Consider the following unbalanced chemical equation. c5h12(l) + o2(g) → co2(g) + h2o(l) if 21.9 gram...
Questions


Health, 30.12.2020 14:00

History, 30.12.2020 14:00

Computers and Technology, 30.12.2020 14:00



Mathematics, 30.12.2020 14:00



Mathematics, 30.12.2020 14:00

Social Studies, 30.12.2020 14:00



Arts, 30.12.2020 14:00

English, 30.12.2020 14:00




History, 30.12.2020 14:00

 = 21.9 g
= 21.9 g = 18 g/mole
= 18 g/mole

 moles of
moles of