

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 01:30
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. the values of phosphorous acid are 1.30 6.70 calculate the ph for each of the given points in the titration of 50.0 ml of 1.5 m h3po3(aq) with 1.5 m koh(aq) .
Answers: 3

Chemistry, 22.06.2019 09:20
What happened to the amount of carbon dioxide in the atmosphere from 2010–2017?
Answers: 1

Chemistry, 22.06.2019 09:30
In 2002, the rare earth elements mine in mountain pass, california was closed because
Answers: 1

You know the right answer?
Ahf [no2] = +33.2 kj mol. use the values below to calculate the standard molar enthalpy change for t...
Questions

Mathematics, 20.09.2019 22:40




Mathematics, 20.09.2019 22:40


History, 20.09.2019 22:40




Computers and Technology, 20.09.2019 22:40

Mathematics, 20.09.2019 22:40

Social Studies, 20.09.2019 22:40


Mathematics, 20.09.2019 22:40

Social Studies, 20.09.2019 22:40

Social Studies, 20.09.2019 22:40

Mathematics, 20.09.2019 22:40


Social Studies, 20.09.2019 22:40

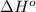
![\Delta H^o_{rxn}=\sum [n\times \Delta H^o_f(product)]-\sum [n\times \Delta H^o_f(reactant)]](/tpl/images/0224/9686/45485.png)
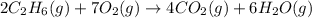
![\Delta H^o_{rxn}=[(4\times \Delta H^o_f_{(CO_2)})+(6\times \Delta H^o_f_{(H_2O)})]-[(2\times \Delta H^o_f_{(C_2H_6)})+(7\times \Delta H^o_f_{(O_2)})]](/tpl/images/0224/9686/92f41.png)
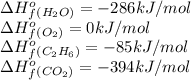
![\Delta H^o_{rxn}=[(4\times (-394))+(6\times (-286))]-[(2\times (-85))+(7\times 0)]\\\\\Delta H^o_{rxn}=-3122kJ](/tpl/images/0224/9686/cf6bf.png)


