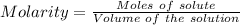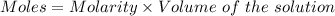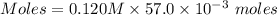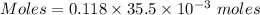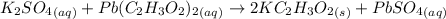Chemistry, 06.09.2019 22:10 loanyst99111
A57.0 ml sample of a 0.120 m potassium sulfate solution is mixed with 35.5 ml of a 0.118 m lead(ii) acetate solution and the following precipitation reaction occurs: k2so4(aq) pb(c2h3o2)2(aq)→2kc2h3o2(aq) pbso4(s) the solid pbso4 is collected, dried, and found to have a mass of 0.992 g . determine the limiting reactant, the theoretical yield, and the percent yield.

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 14:10
13. a covalent bond between two atoms is likely to be polar if: a. one of the atoms is much more electronegative than the other. b. the two atoms are equally electronegative. c. the two atoms are of the same element. d. the bond is part of a tetrahedrally shaped molecule. e. one atom is an anion.
Answers: 1

Chemistry, 22.06.2019 15:40
Use the periodic table to complete this equation that represents nuclear fission processesun - ba c 3 n
Answers: 2

Chemistry, 23.06.2019 10:00
State the effect on the concentration of the clo- ion when there is a decrease in the concentration of the oh- ion
Answers: 1
You know the right answer?
A57.0 ml sample of a 0.120 m potassium sulfate solution is mixed with 35.5 ml of a 0.118 m lead(ii)...
Questions

Mathematics, 13.07.2019 10:30


Chemistry, 13.07.2019 10:30



Mathematics, 13.07.2019 10:30




Advanced Placement (AP), 13.07.2019 10:30



Mathematics, 13.07.2019 10:30


Mathematics, 13.07.2019 10:30


Mathematics, 13.07.2019 10:30



Mathematics, 13.07.2019 10:30