
Chemistry, 06.09.2019 21:30 bludragonfly33
What is the ph of an aqueous solution if the [h") -0.055 m? a) 1.26 b) 2.26 c) 11.74 d) 1.74 e) 6258 e) 0.74

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 00:30
Maria wants to determine which type of disinfectant kills the most bacteria. which of the following is the best way for maria to determine this? a. ask ten different companies that make disinfectants which type is best. b. put the same amount and species of bacteria on ten identical plates, and add ten different kinds of disinfectant to each plate. c. interview ten different people to determine which type of disinfectant they prefer. d. put the same amount and species of bacteria on ten identical plates, and add a different disinfectant to each plate.
Answers: 1

Chemistry, 22.06.2019 08:40
For each of the following compounds, write the formula then predict whether it would be a strong, weak, or non-electrolyte when placed in di water. for the ionic compounds only, put (s) or (aq) after the forrmula formula strong, weak or non electrolyte? a calcium hydroxide b. silver carbonate c. lead(ii) sulfate d. phosphorus trifluoride e. sodium phosphide f barium sulfate g. strontium acetate h. zinc nitrate
Answers: 3

Chemistry, 22.06.2019 12:40
In the following table, all the columns for the element calcium are filled out correctly. element electron structure of atom electron structure of ion net ionic charge calcium 1s22s22p63s23p64s2 1s32s22p63s23p64s1 +1 true false
Answers: 2

Chemistry, 22.06.2019 20:30
From the choices provided below, list the reagent(s) in order that will react with cyclopentanone to form the compound shown below.
Answers: 2
You know the right answer?
What is the ph of an aqueous solution if the [h") -0.055 m? a) 1.26 b) 2.26 c) 11.74 d) 1.74 e) 625...
Questions

English, 28.01.2022 14:00


Mathematics, 28.01.2022 14:00

Mathematics, 28.01.2022 14:00

English, 28.01.2022 14:00


Computers and Technology, 28.01.2022 14:00




Biology, 28.01.2022 14:00



Chemistry, 28.01.2022 14:00


SAT, 28.01.2022 14:00


English, 28.01.2022 14:00


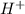 or
or 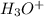 ion in solution.Mathematically it is written as:
ion in solution.Mathematically it is written as: ![pH=-log[H^{+}]=-log[H_{3}O^{+}]](/tpl/images/0224/7325/4dcb4.png) , where
, where ![[H^{+}]or [H_{3}O^{+}]](/tpl/images/0224/7325/2a2e6.png) represents molarity of
represents molarity of 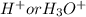 in solution.Here
in solution.Here ![[H^{+}]=0.055 M](/tpl/images/0224/7325/4454d.png) . So,
. So, ![pH=-log[H^{+}]=-log(0.055)=1.26](/tpl/images/0224/7325/056eb.png) pH of the given aqueous solution is 1.26
pH of the given aqueous solution is 1.26

