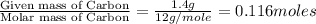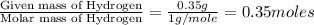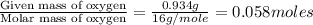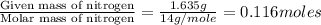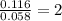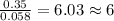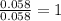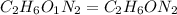Dimethyl nitrosamine is a known carcinogen. it can be formed in the intestinal tract when digestive juices react with the nitrite ion is preserved and smoked meats. it is made up of carbon, hydrogen, nitrogen, and oxygen atoms. a 4.319 g sample of dimethyl nitrosamine burned in oxygen yields 5.134 g of co2 and 3.173 g of h2 o. the compound contains 37,82% by mass of nitrogen. what is the empirical formula of dimethyl nitrosamine?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 19:00
0.66y = 0.9x + 0.48 if y has a value of 108.45 what is the value of x?
Answers: 1

Chemistry, 22.06.2019 09:30
One way that radioactive waste is treated is by burying it in repositories. the repositories are found only in states with very low populations. true or false? a. trueb. false(also i meant to put high school but it put down middle school instead)
Answers: 1

Chemistry, 22.06.2019 22:00
How many moles of no2 will form when 3.3 moles of cu are reacted with excess hno3?
Answers: 3

Chemistry, 23.06.2019 00:30
The molecular weight of carbon dioxide, co2, is 44.00 amu, and the molecular weight of nitrous dioxide, no2, is 46.01 amu, so no2 diffuses co2
Answers: 2
You know the right answer?
Dimethyl nitrosamine is a known carcinogen. it can be formed in the intestinal tract when digestive...
Questions

English, 02.12.2021 01:10


English, 02.12.2021 01:10



Mathematics, 02.12.2021 01:10


Mathematics, 02.12.2021 01:10


Mathematics, 02.12.2021 01:10



History, 02.12.2021 01:10

Biology, 02.12.2021 01:10


Physics, 02.12.2021 01:10

World Languages, 02.12.2021 01:10



Chemistry, 02.12.2021 01:10

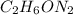
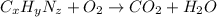
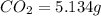
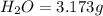
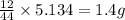 of carbon will be contained.
of carbon will be contained.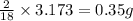 of hydrogen will be contained.
of hydrogen will be contained.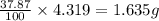 of nitrogen
of nitrogen