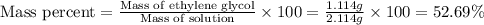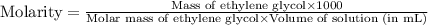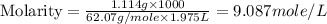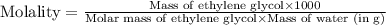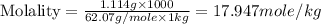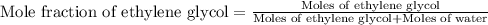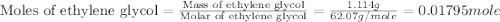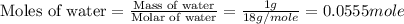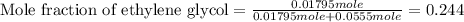An automobile antifreeze mixture is made by mixing equal volumes of ethylene glycol (d = 1.114 g/ml; m = 62.07 g/mol) and water (d = 1.00 g/ml) at 20°c. the density of the mixture is 1.070 g/ml. express the concentration of ethylene glycol as
a- volume percent
b- mass percent
c- molarity
d- molality
e- mole fraction

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 10:00
Diffraction is when light is bent around obstructions. which of the these observation about clouds would indicate diffraction? a) after rain storms, you can sometimes see rainbows. b) clouds are white or gray and cannot be seen through. c) on a cloudy day, the temperature tends to be cooler than a sunny day. d) the edges of dark clouds appear lighter. this
Answers: 3


Chemistry, 23.06.2019 05:00
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 4.20 mol fe and 6.70 mol nio(oh) react?
Answers: 3

You know the right answer?
An automobile antifreeze mixture is made by mixing equal volumes of ethylene glycol (d = 1.114 g/ml;...
Questions



History, 20.04.2021 03:30

Mathematics, 20.04.2021 03:30

Physics, 20.04.2021 03:30

Mathematics, 20.04.2021 03:30

Mathematics, 20.04.2021 03:30

English, 20.04.2021 03:30



Mathematics, 20.04.2021 03:30




Mathematics, 20.04.2021 03:30



Mathematics, 20.04.2021 03:30

Mathematics, 20.04.2021 03:30

Mathematics, 20.04.2021 03:30