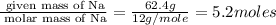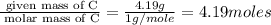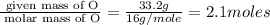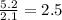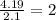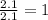Chemistry, 05.09.2019 17:10 samsmith666
Measurements show that unknown compound x has the following composition:
element mass %
carbon 62.4
hydrogen 4.19
oxygen 33.2
write the empirical chemical formula of x?

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 10:00
Ahydrogen atom has 1 electron. how many bonds can hydrogen form? a) 1 b) 2 c) 3 d) 4 e) 5
Answers: 3

Chemistry, 22.06.2019 13:00
6. using 3 – 4 sentences explain (in your own words) why water expands when it freezes? 7. using your knowledge of colligative properties explain whether sodium chloride or calcium chloride would be a more effective substance to melt the ice on a slick sidewalk. use 3 – 4 sentences in your explanation.
Answers: 1

Chemistry, 22.06.2019 17:10
)benzene and toluene form nearly ideal solutions. consider an equimolar solution of benzene and toluene. at 20 °c the vapour pressures of pure benzene and toluene are 9.9 kpa and 2.9 kpa, respectively. the solution is boiled by reducing the external pressure below the vapour pressure. calculate (i) the pressure when boiling begins, (ii) the composition of each component in the vapour, and (iii) the vapour pressure when only a few drops of liquid remain. assume that the rate of vaporization is low enough for the temperature to remain constant at 20 °c.
Answers: 1
You know the right answer?
Measurements show that unknown compound x has the following composition:
element mass %
...
element mass %
...
Questions



Mathematics, 23.04.2020 19:45



Social Studies, 23.04.2020 19:45



Chemistry, 23.04.2020 19:45

Mathematics, 23.04.2020 19:45






Chemistry, 23.04.2020 19:45


Social Studies, 23.04.2020 19:45

Spanish, 23.04.2020 19:45

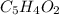 .
.