
Chemistry, 05.09.2019 02:30 xboxdude06
[cu(nh3)4]2+ solutions exhibit a deep blue-violet color. how can you use spectrophotometry to confirm that this reaction has occurred and that the product formed is tetraamminecopper (ii) sulfate?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 15:00
Many ionic compounds and a few highly polar covalent compounds are because they completely ionize in water to create a solution filled with charged ions that can conduct an electric current.
Answers: 1

Chemistry, 22.06.2019 20:00
What happens to the temperature of a substance when the average kinetic energy of its particles increases?
Answers: 3

Chemistry, 22.06.2019 21:30
Liquid ammonia is produced at high temperatures and under great pressure in a tank by passing a mixture of nitrogen gas and hydrogen gas over an iron catalyst. the reaction is represented by the following equation. n2(g) + 3h2(g) → 2nh3(g) changing all but one experimental condition will affect the amount of ammonia produced. that condition is a) increasing the concentration of both reactants b) changing the temperature within the tank c) decreasing the pressure within the tank. d) increasing only the amount of nitrogen present.
Answers: 1

Chemistry, 23.06.2019 15:00
Solve this problem using the appropriate law. (remember that ) what is the pressure of 1.9 mols of nitrogen gas in a 9.45 l tank and at a temperature of 228 k?
Answers: 1
You know the right answer?
[cu(nh3)4]2+ solutions exhibit a deep blue-violet color. how can you use spectrophotometry to confir...
Questions


History, 19.03.2020 03:13


Computers and Technology, 19.03.2020 03:14



Computers and Technology, 19.03.2020 03:14




Biology, 19.03.2020 03:15

Spanish, 19.03.2020 03:15



English, 19.03.2020 03:15



Mathematics, 19.03.2020 03:15

History, 19.03.2020 03:16

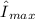 ), which can be looked up in literature. This characteristic λmax is different from the characteristic λmax of the reagents, as [Cu(NH₃)₄]²⁺ absorbs red-orange light and thus appears with a deep blue-violet color.
), which can be looked up in literature. This characteristic λmax is different from the characteristic λmax of the reagents, as [Cu(NH₃)₄]²⁺ absorbs red-orange light and thus appears with a deep blue-violet color.

