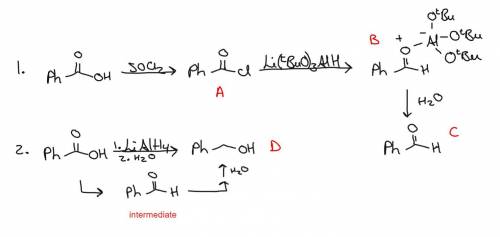
Two students are given the starting material benzoic acid and are asked to prepare benzaldehyde. the first student starts by refluxing her sample of benzoic acid in thionyl chloride in the fume hood. upon completion of the reaction, she evaporates the thionyl chloride to isolate compound a. she treats compound a with a stoichiometric amount of lithium tri-tert-butoxyaluminum hydride at –78 °c in diethyl ether, producing compound b. adding water, she isolates her product, compound c. the second student takes a different route. she treats benzoic acid with an excess of lithium aluminum hydride (lah) in diethyl ether, followed by careful addition of ethyl acetate to remove any unreacted lah. she adds water and isolates her product, compound d. draw the structure for compound a, compound c, and compound d below.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 13:30
Forests and meadows are often cut down to make way for farms or large number of new homes. what are some of the elements of ecosystems that are lost when plants in these areas are removed?
Answers: 2

Chemistry, 22.06.2019 02:30
You have a sample of a gas that occupies a volume of 17ml at -111 degrees celsius. what volume does the sample occupy at 88 degrees celsius? show all work asap
Answers: 3

Chemistry, 22.06.2019 06:30
If 1.8 l of water is added to 2.5l of a 7.0 m koh solution, what is the molarity of the new solution
Answers: 1

Chemistry, 22.06.2019 11:00
Predict the products of the following acid-base reactions, and predict whether the equilibrium lies to the left or to the right of the reaction arrow.part ao2-(aq)+h2o(l)< => express your answer as part of a chemical equation. identify all of the phases in your answer.o2-(aq)+h2o(l) < => oh-(aq)+oh-(aq)part bpredict whether the equilibrium lies to the left or to the right of the equation in previous part.h2o is a stronger acid than oh–, so the equilibrium lies to the right.h2o is a weaker acid than oh–, so the equilibrium lies to the left.h2o is a stronger acid than oh–, so the equilibrium lies to the left.h2o is a weaker acid than oh–, so the equilibrium lies to the right.part cch3cooh(aq)+hs? (aq) < => express your answer as part of a chemical equation. identify all of the phases in your answer.ch3cooh(aq)+hs-(aq) < => h2s(aq)+c2h3o2-(aq)h2s(aq)+c2h3o2-(aq)part dpredict whether the equilibrium lies to the left or to the right of the equation in previous part.ch3cooh is a weaker acid than h2s, so the equilibrium lies to the right.ch3cooh is a weaker acid than h2s, so the equilibrium lies to the left.ch3cooh is a stronger acid than h2s, so the equilibrium lies to the right.ch3cooh is a stronger acid than h2s, so the equilibrium lies to the left.part eno2-(aq)+h2o(l) < => express your answer as part of a chemical equation. identify all of the phases in your answer.no2-(aq)+h2o(l) < => part fpredict whether the equilibrium lies to the left or to the right of the equation in previous part.hno2 is a stronger acid than h2o, so the equilibrium lies to the right.hno2 is a weaker acid than h2o, so the equilibrium lies to the left.hno2 is a stronger acid than h2o, so the equilibrium lies to the left.hno2 is a weaker acid than h2o, so the equilibrium lies to the right.
Answers: 1
You know the right answer?
Two students are given the starting material benzoic acid and are asked to prepare benzaldehyde. the...
Questions

Mathematics, 15.11.2021 01:00




Mathematics, 15.11.2021 01:00


Mathematics, 15.11.2021 01:00

History, 15.11.2021 01:00

Mathematics, 15.11.2021 01:00




Mathematics, 15.11.2021 01:00

Mathematics, 15.11.2021 01:00

Mathematics, 15.11.2021 01:00

Mathematics, 15.11.2021 01:00