
Chemistry, 04.09.2019 02:30 peytonbrien2002
Pure magnesium metal is often found as ribbons and can easily burn in the presence of oxygen. when 4.51 g of magnesium ribbon burns with 6.92 g of oxygen, a bright, white light and a white, powdery product are formed. enter the balanced chemical equation for this reaction. be sure to include all physical states.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 12:50
The number at the end of an isotope’s name is the number.
Answers: 1

Chemistry, 22.06.2019 15:30
A1.5l container holds p.50 grams of an unknown gas at a pressure of 0.44 atm and a temperature of 50.c what is the molar mass of the unknown gas
Answers: 1

Chemistry, 22.06.2019 17:40
Which statement about hf is true? it is zero for any compound in its standard state. it is positive when the bonds of the product store more energy than those of the reactants. it is negative when a compound forms from elements in their standard states. it is zero for any element that is in the liquid state.
Answers: 1

Chemistry, 22.06.2019 19:50
A2.5% (by mass) solution concentration signifies that there is a 2.5 % (by mass) solution concentration signifies that there is blank of solute in every 100 g of solution. of solute in every 100 g of solution
Answers: 3
You know the right answer?
Pure magnesium metal is often found as ribbons and can easily burn in the presence of oxygen. when 4...
Questions


Mathematics, 22.04.2021 07:00

Biology, 22.04.2021 07:00



Mathematics, 22.04.2021 07:00




Mathematics, 22.04.2021 07:00


Mathematics, 22.04.2021 07:00

History, 22.04.2021 07:00

Mathematics, 22.04.2021 07:00



History, 22.04.2021 07:00

History, 22.04.2021 07:00

Biology, 22.04.2021 07:00

Computers and Technology, 22.04.2021 07:00

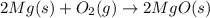
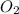 is present in gaseous state, and MgO is present in solid state.
is present in gaseous state, and MgO is present in solid state.

