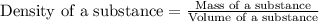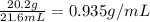Chemistry, 03.09.2019 23:30 iibabycarrotsii
To save time you can approximate the initial volume of water to ±1 ml and the initial mass of the solid to ±1 g. for example, if you are asked to add 23 ml of water, add between 22 ml and 24 ml. which metals in each of the following sets will have equal density? *20.2 g gold placed in 21.6 ml of water and 12.0 g copper placed in 21.6 ml of water. *20.2 g silver placed in 21.6 ml of water and 12.0 g silver placed in 21.6 ml of water. *15.2 g copper placed in 21.6 ml of water and 50.0 g copper placed in 23.4 ml of water. *15.4 g gold placed in 20.0 ml of water and 15.7 g silver placed in 20.0 ml of water. *20.2 g silver placed in 21.6 ml of water and 20.2 g copper placed in 21.6 ml of water. *11.2 g gold placed in 21.6 ml of water and 14.9 g gold placed in 23.4 ml of water.

Answers: 3
Another question on Chemistry



Chemistry, 23.06.2019 06:20
An object of mass 10.0 kg and volume 1000 ml and density 10 g/ml sinks in water who’s density is 1.0 g/ml. what is the mass of the water which has been displaced in kilograms
Answers: 1

Chemistry, 23.06.2019 10:00
The temperature of a lead fishing weight rises from 26 °c to 38 °c as it absorbs 11.3 j of heat. what is the mass of the fishing weight in grams?
Answers: 2
You know the right answer?
To save time you can approximate the initial volume of water to ±1 ml and the initial mass of the so...
Questions

English, 03.03.2021 21:00

Geography, 03.03.2021 21:00

Social Studies, 03.03.2021 21:00

English, 03.03.2021 21:00

Mathematics, 03.03.2021 21:00



Mathematics, 03.03.2021 21:00




Mathematics, 03.03.2021 21:00


Mathematics, 03.03.2021 21:00


Mathematics, 03.03.2021 21:00


Mathematics, 03.03.2021 21:00

Mathematics, 03.03.2021 21:00