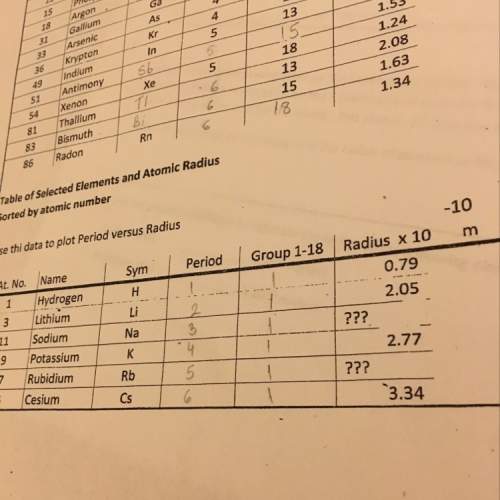
The mass of MgO formed is 3.502 grams and mass of 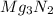 formed is 1.587 grams.
formed is 1.587 grams.
Explanation:
We are given:
Total mass of magnesium reacted = 3.26 g
Total mass of products formed = 5.09 g
To calculate number of moles of a substance, we use the equation:
 .....(1)
.....(1)
For calculating mass of MgO:
The chemical equation for the formation of magnesium oxide follows:
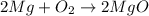
Let 'x' grams of magnesium is reacting to form magnesium oxide.
Molar mass of Magnesium = 24.3 g/mol
Putting values in equation 1, we get:
Number of moles of magnesium = 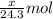
By Stoichiometry of the reaction:
2 moles of magnesium produces 2 moles of MgO
So, 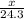 moles of magnesium will produce
moles of magnesium will produce 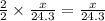 moles of MgO
moles of MgO
Molar mass of MgO = 40.3 g/mol
Putting values in equation 1, we get:
Mass of MgO = 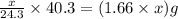
Mass of magnesium oxide = 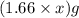 = X grams
= X grams
For calculating mass of
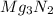
:
The chemical equation for the formation of magnesium nitride follows:
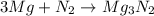
Mass of magnesium reacting = (3.26 - x)
Molar mass of Magnesium = 24.3 g/mol
Putting values in equation 1, we get:
Number of moles of magnesium = 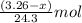
By Stoichiometry of the reaction:
3 moles of magnesium produces 1 moles of 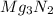
So, 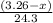 moles of magnesium will produce
moles of magnesium will produce  moles of
moles of 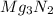
Molar mass of 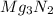 = 101 g/mol
= 101 g/mol
Putting values in equation 1, we get:
Mass of ![Mg_3N_2=\frac{(3.26-x)}{72.9}\times 101=[(3.26-x)\times 1.38]g](/tpl/images/0221/4247/e57b9.png)
Mass of magnesium nitride = ![[(3.26-x)\times 1.38]g](/tpl/images/0221/4247/79581.png) = Y grams
= Y grams
Calculating the mass of products:
Total mass of the products = 5.09 grams
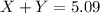
Putting values of 'X' and 'Y' in above equation, we get:
![(1.66\times x)+[(3.26-x)\times 1.38]=5.09\\\\x=2.11g](/tpl/images/0221/4247/08016.png)
Mass of MgO = 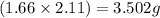
Mass of ![Mg_3N_2=[(3.26-2.11)\times 1.38]=1.587g](/tpl/images/0221/4247/42984.png)
Hence, the mass of MgO formed is 3.502 grams and mass of 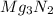 formed is 1.587 grams.
formed is 1.587 grams.



























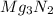 formed is 1.587 grams.
formed is 1.587 grams. .....(1)
.....(1)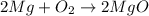
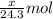
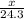 moles of magnesium will produce
moles of magnesium will produce 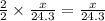 moles of MgO
moles of MgO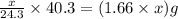
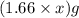 = X grams
= X grams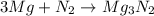
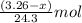
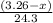 moles of magnesium will produce
moles of magnesium will produce  moles of
moles of ![Mg_3N_2=\frac{(3.26-x)}{72.9}\times 101=[(3.26-x)\times 1.38]g](/tpl/images/0221/4247/e57b9.png)
![[(3.26-x)\times 1.38]g](/tpl/images/0221/4247/79581.png) = Y grams
= Y grams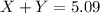
![(1.66\times x)+[(3.26-x)\times 1.38]=5.09\\\\x=2.11g](/tpl/images/0221/4247/08016.png)
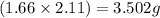
![Mg_3N_2=[(3.26-2.11)\times 1.38]=1.587g](/tpl/images/0221/4247/42984.png)