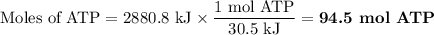Chemistry, 31.08.2019 03:10 darianhaynes
A) the metabolism of glucose can be written as c6h12o6(s) + 602() - → 6co2(8) + 6h20m calculate the value of ahⓡ and aso for the reaction b) calculate the value of agº for the reaction at 298 k. c) cells use energy derived from the metabolism of glucose to synthesize atp from adp and phosphate ion(written as p). adp + p → atp that reaction has agⓡ = +30.5 kj/mol. how many moles of atp could theoretically be made from the metabolism of 1 mole of glucose?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 23:40
If the atomic mass of an atom is 34 and the atom contains 13 protons, how many neutrons does the atom contain?
Answers: 2

Chemistry, 22.06.2019 16:30
How many grams of mgbr2 are needed to produce 75g or metal?
Answers: 1

Chemistry, 22.06.2019 20:00
Phosphoric acid is a triprotic acid ( =6.9×10−3 , =6.2×10−8 , and =4.8×10−13 ). to find the ph of a buffer composed of h2po−4(aq) and hpo2−4(aq) , which p value should be used in the henderson–hasselbalch equation? p k a1 = 2.16 p k a2 = 7.21 p k a3 = 12.32 calculate the ph of a buffer solution obtained by dissolving 18.0 18.0 g of kh2po4(s) kh 2 po 4 ( s ) and 33.0 33.0 g of na2hpo4(s) na 2 hpo 4 ( s ) in water and then diluting to 1.00 l.
Answers: 3

You know the right answer?
A) the metabolism of glucose can be written as c6h12o6(s) + 602() - → 6co2(8) + 6h20m calculate the...
Questions



Mathematics, 27.06.2020 20:01



English, 27.06.2020 20:01










Mathematics, 27.06.2020 20:01