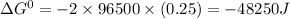11. (12pts) for the redox reaction given below, ni(s) + 2 h(aq) + ni2+(aq) + h2(e) (show your work in detail) a) write oxidation and reduction half cell reactions b) write the electrochemical cell notation c) predict whether this reaction will be spontaneous. explain. d) calculategº 6 page

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 23:00
What is the molecular formula for a compound that is 46.16% carbon, 5.16% hydrogen, and 48.68% fluorine? the molar mass of the compound is 156.12 g/mol
Answers: 2

Chemistry, 22.06.2019 02:30
When you perform this reaction, what could remain at the end of the reaction? check all that apply. excess reactant aqueous copper chloride excess reactant aluminum oxygen product solid copper carbon dioxide product aqueous aluminum chloride water
Answers: 2

Chemistry, 22.06.2019 07:00
How far is the region from the equator or control climate
Answers: 1

Chemistry, 22.06.2019 07:30
Given that 1 mi = 1760 yd, determine what conver- sion factor is appropriate to convert 1849 yd to miles; to convert 2.781 mi to yards.
Answers: 2
You know the right answer?
11. (12pts) for the redox reaction given below, ni(s) + 2 h(aq) + ni2+(aq) + h2(e) (show your work i...
Questions


Mathematics, 26.05.2021 19:50



Mathematics, 26.05.2021 19:50


Mathematics, 26.05.2021 19:50

Arts, 26.05.2021 19:50



Medicine, 26.05.2021 19:50

Mathematics, 26.05.2021 19:50

Mathematics, 26.05.2021 19:50

English, 26.05.2021 19:50

Social Studies, 26.05.2021 19:50

Mathematics, 26.05.2021 19:50

Biology, 26.05.2021 19:50



Geography, 26.05.2021 19:50

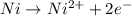
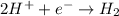
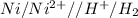
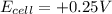 , the reaction is spontaneous.
, the reaction is spontaneous.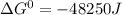
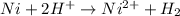
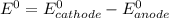
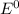 are standard reduction potentials.
are standard reduction potentials.![E^0_{[Ni^{2+}/Ni]}= -0.25V](/tpl/images/0213/4556/864ce.png)
![E^0_{[H^{+}/H_2]}=+0.0V](/tpl/images/0213/4556/826b0.png)
![E^0=E^0_{[H^{+}/H_2]}- E^0_{[Ni^{2+}/Ni]}](/tpl/images/0213/4556/3d59c.png)
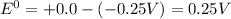
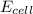 = +ve, reaction is spontaneous
= +ve, reaction is spontaneous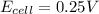 , the reaction is spontaneous.
, the reaction is spontaneous.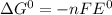
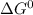 = standard gibbs free energy
= standard gibbs free energy