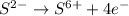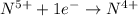Consider the following redox reaction: (38) cus + no3 → cuso4 + no2 which of the following statement is false? [1] [2] [3] cu has an oxidation state of +2 in cus. cu has an oxidation state of +2 in cuso4. s is oxidized in the above reaction. electrons are donated from s in cus to n in no3". none of the above.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 10:30
What woukd most likely be the transmittance at a 0.70 m solution of solute a? a) 7.6%b) 1.1%c)4.0%d)4.6%
Answers: 1


Chemistry, 22.06.2019 20:30
How many grams of phosphorus are contained in 5.09 moles of phosphorus?
Answers: 1

Chemistry, 22.06.2019 23:00
If two identical atoms are bonded,what kind of molecule is formed
Answers: 1
You know the right answer?
Consider the following redox reaction: (38) cus + no3 → cuso4 + no2 which of the following statemen...
Questions






Computers and Technology, 10.12.2019 19:31









Computers and Technology, 10.12.2019 19:31





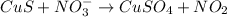
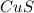 and
and 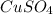 is, (+2).
is, (+2).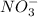 .
.