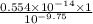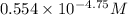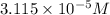Chemistry, 30.08.2019 03:00 apreston2882
You add 30ml of 1m of nh3 to a 50ml solution of 0,2 mgcl2. magnesium hydroxide (mg(oh)2) precipitates.
what is the minimum concentration of nh4+ that prevents the precipitation of mg(oh)2?
given information:
(mgoh2) ksp = 13
(nh3) kb = 10^-4.75
note: a complex formation between mg and nh4+ is ruled out.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 03:00
In the 1800s, one of the statements in john dalton's atomic theory was that atoms are indivisible. later experimental evidence led to the discovery of subatomic particles such as neutrons, electrons, and protons. what happened to the indivisible atom part of dalton's atomic theory, and why?
Answers: 3

Chemistry, 22.06.2019 12:00
An atom of which element reacts with an atom of hydrogen to form a bond with the greatest degree of polarity ?
Answers: 1

Chemistry, 22.06.2019 12:10
Achemistry student needs to standardize a fresh solution of sodium hydroxide. he carefully weighs out of oxalic acid , a diprotic acid that can be purchased inexpensively in high purity, and dissolves it in of distilled water. the student then titrates the oxalic acid solution with his sodium hydroxide solution. when the titration reaches the equivalence point, the student finds he has used of sodium hydroxide solution.calculate the molarity of the student's sodium hydroxide solution. be sure your answer has the correct number of significant digits.
Answers: 1

Chemistry, 22.06.2019 17:30
To find the enthalpy of a reaction in the lab, you measured the of the reactants and the change during the reaction.
Answers: 1
You know the right answer?
You add 30ml of 1m of nh3 to a 50ml solution of 0,2 mgcl2. magnesium hydroxide (mg(oh)2) precipitate...
Questions

English, 12.10.2020 21:01

Computers and Technology, 12.10.2020 21:01



Social Studies, 12.10.2020 21:01

Spanish, 12.10.2020 21:01

Mathematics, 12.10.2020 21:01

Mathematics, 12.10.2020 21:01





History, 12.10.2020 21:01

Mathematics, 12.10.2020 21:01

Mathematics, 12.10.2020 21:01

Mathematics, 12.10.2020 21:01




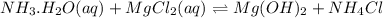
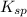 of
of 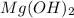 = 13 and
= 13 and 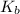 of
of 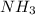 =
= 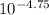
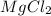 =
= 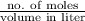
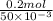
![Mg(OH)_{2} \rightleftharpoons Mg^{2+} + 2[OH^{-}]](/tpl/images/0210/7309/b229d.png)
![K_{sp} = [Mg^{2+}][OH^{-}]^{2}](/tpl/images/0210/7309/b67d6.png)
![4 \times [OH^{-}]^{2}](/tpl/images/0210/7309/cb444.png)
![[OH^{-}]](/tpl/images/0210/7309/e46dd.png) = 1.80276 M
= 1.80276 M![K_{w} = [H_{3}O^{+}][OH^{-}]](/tpl/images/0210/7309/912a0.png) ......... (1)
......... (1)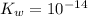
![[OH^{-}] = \frac{K_{w}}{[H_{3}O^{+}]}](/tpl/images/0210/7309/47cf2.png)
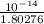
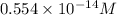
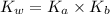 =
= 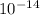
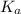 =
= 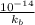
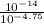
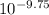
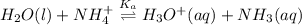
![K_{a} = \frac{[H_{3}O^{+}[NH_{3}]]}{[NH^{+}_{4}]}](/tpl/images/0210/7309/251f5.png) ....... (2)
....... (2)![[NH^{+}_{4}]](/tpl/images/0210/7309/286e5.png) as follows.
as follows.![\frac{[H_{3}O^{+}][NH_{3}]}{K_{a}}](/tpl/images/0210/7309/e4886.png)