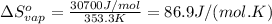Chemistry, 30.08.2019 01:30 veneciaconton347
The enthalpy of vaporization (δh°vap) of benzene is 30.7 kj/mol at its normal boiling point of 353.3 k. what is δs°vap at this temperature? a. 383 j/(mol·k) b. 0.0115 j/(mol·k) c. 86.9 j/(mol·k) d. 0.087 j/(mol·k) e. 11.5 j/(mol·k)

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 18:30
What volume of a 2.00 m stock solution of naoh is needed to prepare 150. ml of 0.40 m solution?
Answers: 2

Chemistry, 22.06.2019 13:00
12. calculate the hydroxide ion concentration of a solution with ph = 3.25. show all calculations leading to an answer
Answers: 3

Chemistry, 22.06.2019 23:30
If it is an isoelectronic series select true, if not select false. o2-, s2-, se2-, te2- na+, k+, rb+, cs+ n3-, p3-, as3-, sb3- ag, cd+, sn3+, sb4+ f-, cl-, br-, i- f-, ne, na+, mg2+ s2-, s, s6+
Answers: 1

Chemistry, 23.06.2019 01:00
Which of the following is a physical change? a.burning a piece of wood b.sawing a piece of wood in half c.rust forming on an iron fence d.a copper roof changing color from orange to green
Answers: 1
You know the right answer?
The enthalpy of vaporization (δh°vap) of benzene is 30.7 kj/mol at its normal boiling point of 353.3...
Questions



Social Studies, 12.11.2020 07:00

Mathematics, 12.11.2020 07:00

History, 12.11.2020 07:00

Mathematics, 12.11.2020 07:00


History, 12.11.2020 07:00

Health, 12.11.2020 07:00

Advanced Placement (AP), 12.11.2020 07:00

Mathematics, 12.11.2020 07:00

Business, 12.11.2020 07:00

Spanish, 12.11.2020 07:00

Mathematics, 12.11.2020 07:00

Mathematics, 12.11.2020 07:00


Mathematics, 12.11.2020 07:00

Mathematics, 12.11.2020 07:00

Mathematics, 12.11.2020 07:00

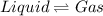
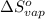 for the reaction, we use the equation:
for the reaction, we use the equation: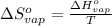
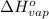 = standard enthalpy change of vaporization = 30.7 kJ/mol = 30700 J/mol (Conversion factor: 1 kJ = 1000 J)
= standard enthalpy change of vaporization = 30.7 kJ/mol = 30700 J/mol (Conversion factor: 1 kJ = 1000 J)