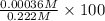Chemistry, 29.08.2019 18:00 lhmsokol56
Calculate the percent ionization of nitrous acid in a solution that is 0.222 m in nitrous acid (hno3) and 0.278 m in potassium nitrite (kno2). the acid dissociation constant of nitrous acid is 4.50 x 10-4 a) 55.6 b) 0.162 c) 15.5 d) 2.78 * 10-3

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 20:30
There is an area in idaho named craters of the moon where most of the ground is covered with basalt, adark gray, igneous rock with no visibl crystals. what can you infer about the geographical history of the area?
Answers: 1

Chemistry, 22.06.2019 06:30
Three cards with holes are arranged in a straight line. a light is shined through the first card’s hole and travels through all three cards. what does this tell you about light rays? a) that light is reflected b) that light is refractive c) that light travels in a straight line d) that light does not travel in a straight line
Answers: 1

Chemistry, 22.06.2019 07:00
Achemist wants to extract copper metal from copper chloride solution. the chemist places 0.50 grams of aluminum foil in a solution containing 0.75 grams of copper (ii) chloride. a single replacement reaction takes place. (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction? a) approximately 0.36 grams, because copper (ii) chloride acts as a limiting reactant b) approximately 1.8 grams, because copper (ii) chloride acts as a limiting reactant c) approximately 0.36 grams, because aluminum acts as a limiting reactant d) approximately 1.8 grams, because aluminum acts as a limiting reactant
Answers: 3

You know the right answer?
Calculate the percent ionization of nitrous acid in a solution that is 0.222 m in nitrous acid (hno3...
Questions




Mathematics, 05.05.2020 19:34


Social Studies, 05.05.2020 19:34

Mathematics, 05.05.2020 19:34

History, 05.05.2020 19:34

Mathematics, 05.05.2020 19:34


History, 05.05.2020 19:34




Mathematics, 05.05.2020 19:34


History, 05.05.2020 19:34


Mathematics, 05.05.2020 19:34

English, 05.05.2020 19:34

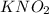 is 0.278 M and it is completely ionized into
is 0.278 M and it is completely ionized into 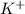 and
and 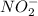 .
.![[KNO_{2}]](/tpl/images/0209/3896/fb191.png) =
= ![[NO_{2}]](/tpl/images/0209/3896/53e25.png) = 0.278 M
= 0.278 M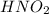 is 0.222 M.
is 0.222 M.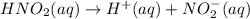
![K_{a} = \frac{[H^{+}][NO^{-}_{2}]}{[HNO_{2}]}](/tpl/images/0209/3896/218b7.png)
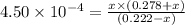
![[H^{+}]](/tpl/images/0209/3896/85507.png) is 0.00036 M. Therefore, percentage ionization of
is 0.00036 M. Therefore, percentage ionization of ![\frac{[H^{+}]}{[HNO_{2}]} \times 100](/tpl/images/0209/3896/26d17.png)