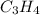Chemistry, 28.08.2019 21:10 heavenwagner
Cumene is a compound containing only carbon and hydrogen that is used in the production of acetone and phenol in the chemical industry. combustion of 47.6 mg cumene produces some co2 and 42.8 mg water. the molar mass of cumene is between 115 and 125 g/mol. determine the empirical and molecular formulas.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 20:30
The balanced chemical equation for this lab is: 3cucl2(aq) + 2al(s) 3cu(s) + 2alcl3(aq) if 10.5 g copper chloride react with 12.4 g aluminum, what is the limiting reactant?
Answers: 3



Chemistry, 23.06.2019 00:00
The empirical formula of a compound is ch2o and its mass is 120 amu/molecule, what is its formula?
Answers: 2
You know the right answer?
Cumene is a compound containing only carbon and hydrogen that is used in the production of acetone a...
Questions

Mathematics, 02.03.2021 21:20

Mathematics, 02.03.2021 21:20

Biology, 02.03.2021 21:20

Chemistry, 02.03.2021 21:20

History, 02.03.2021 21:20




Geography, 02.03.2021 21:20



Physics, 02.03.2021 21:20

Mathematics, 02.03.2021 21:20

Chemistry, 02.03.2021 21:20

Mathematics, 02.03.2021 21:20

Chemistry, 02.03.2021 21:20




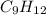
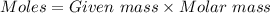
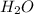 = 42.8/18 = 2.3778×10⁻³ moles
= 42.8/18 = 2.3778×10⁻³ moles