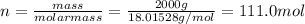Chemistry, 27.08.2019 17:10 Jackiebear4593
Assuming the mass of the ice in the beaker was 2000 g (2 kg), calculate the amount of heat that must have been added to convert the ice to water (assume that the entire 2000 g was ice at the start of melting, and the entire 2000 g was water at the end of the melting). assume that the change took place entirely at 0 °c. the heat of fusion (dhfus) for h2o is 6.12 kj/mol.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 05:00
When you mate two plants together the terms is called? answer it fast as possible plz! i have a test tomorrow!
Answers: 1

Chemistry, 22.06.2019 05:30
Which other elements contain the same number of outer electrons as sodium
Answers: 3

Chemistry, 22.06.2019 13:00
Is 9 correct? and can someone me with 10? it’s due tomorrow, you
Answers: 1

You know the right answer?
Assuming the mass of the ice in the beaker was 2000 g (2 kg), calculate the amount of heat that must...
Questions

English, 12.12.2019 15:31

Social Studies, 12.12.2019 15:31


Mathematics, 12.12.2019 16:31


History, 12.12.2019 16:31

Mathematics, 12.12.2019 16:31


English, 12.12.2019 16:31

Mathematics, 12.12.2019 16:31

Mathematics, 12.12.2019 16:31


Chemistry, 12.12.2019 16:31



Mathematics, 12.12.2019 16:31

Mathematics, 12.12.2019 16:31

English, 12.12.2019 16:31