
Chemistry, 27.08.2019 16:30 brandy2115
Na2co3 + hcl → co2 + h2o + naclconsider the above unbalanced equation. for this reaction, how many ml of a 2 m solution of na2co3 are required to produce 11.2 l of co2 at stp?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 17:10
A+b→2c when the reaction begins, the researcher records that the rate of reaction is such that 1 mole of a is consumed per minute. after making changes to the reaction, the researcher notes that 2 moles of a are consumed per minute. what change could the researcher have made to effect this change?
Answers: 1

Chemistry, 22.06.2019 01:40
Darla claims that the first periodic table developed by mendeleev was not completely accurate, so it is not useful at all. harmony argues that it establish the periodic table we use today, making it more credible. who is correct and why? darla is correct, because a model that has any mistakes should be thrown out. darla is correct, because a good model would not need to change. harmony is correct, because mendeleev’s model had all of the information correct in the first version. harmony is correct, because mendeleev’s model made predictions that came true.
Answers: 1

Chemistry, 22.06.2019 07:40
21. consider the following chemical reaction: n2+ o2 2 no if 10.0 g of n2 reacts with excess oxygen then how many grams of no can be formed? a) 10.7 g b) 21.4 g c) 32.9 g d) 42.8 g page 4 of 8
Answers: 2

Chemistry, 22.06.2019 14:30
Consider the reduction reactions and their equilibrium constants. cu+(aq)+e−↽−−⇀cu(s)pb2+(aq)+2e−↽−−⇀pb(s)fe3+(aq)+3e−↽−−⇀fe(=6.2×108=4.0×10−5=9.3×10−3 cu + ( aq ) + e − ↽ − − ⇀ cu ( s ) k =6.2× 10 8 pb 2 + ( aq ) +2 e − ↽ − − ⇀ pb ( s ) k =4.0× 10 − 5 fe 3 + ( aq ) +3 e − ↽ − − ⇀ fe ( s ) k =9.3× 10 − 3 arrange these ions from strongest to weakest oxidizing agent.
Answers: 3
You know the right answer?
Na2co3 + hcl → co2 + h2o + naclconsider the above unbalanced equation. for this reaction, how many m...
Questions


Geography, 27.01.2020 20:31

Chemistry, 27.01.2020 20:31



Spanish, 27.01.2020 20:31



Mathematics, 27.01.2020 20:31

History, 27.01.2020 20:31

Mathematics, 27.01.2020 20:31



Mathematics, 27.01.2020 20:31

Social Studies, 27.01.2020 20:31

Biology, 27.01.2020 20:31




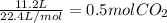
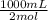 = 250 mL
= 250 mL

