
Sulfur dioxide gas reacts with sodium hydroxide to form sodium sulfite and water. the unbalanced chemical equation for this reaction is given below: so2(g) + naoh(s) → na2so3(s) + h2o(l)assuming that you start with 36.8 g of sulfur dioxide and 20.7 g of sodium hydroxide and assuming that the reaction goes to completion, determine the mass of each product formed. g na2so3g h2o

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 15:10
Atank contains 240 liters of fluid in which 10 grams of salt is dissolved. brine containing 1 gram of salt per liter is then pumped into the tank at a rate of 6 l/min; the well-mixed solution is pumped out at the same rate. find the number a(t) of grams of salt in the tank at time t.
Answers: 3

Chemistry, 22.06.2019 16:00
If 15 drops of ethanol from a medical dropper weight 0.60g, how many drops does it takes from a dropper to dispense 1.0ml of ethanol? the density of ethanol is 0.80g/ml
Answers: 1

Chemistry, 23.06.2019 02:00
Which best describes the present-day universe? opaque, expanding very slowly, stars produce heavy elements transparent, expanding at an accelerated rate, stars produce heavy elements opaque, expanding at an accelerated rate, stars produce only hydrogen and helium transparent, expanding very slowly, stars produce only hydrogen and helium
Answers: 1

Chemistry, 23.06.2019 10:10
Calculate the h3o+ concentration in a solution of acetic acid if the concentration of molecular acetic acid present at equilibrium is 9.97x10-3 m and k for the dissociation is 1.86x10-5. ch3cooh(aq)+h2o(l)+> h3o+(aq)+ch3coo-(aq) show me how to get the answer.
Answers: 3
You know the right answer?
Sulfur dioxide gas reacts with sodium hydroxide to form sodium sulfite and water. the unbalanced che...
Questions


Mathematics, 02.12.2020 19:50

Biology, 02.12.2020 19:50


History, 02.12.2020 19:50

Biology, 02.12.2020 19:50


Geography, 02.12.2020 19:50

Arts, 02.12.2020 19:50


English, 02.12.2020 19:50

Mathematics, 02.12.2020 19:50

English, 02.12.2020 19:50

Mathematics, 02.12.2020 19:50




History, 02.12.2020 19:50

Business, 02.12.2020 19:50

Mathematics, 02.12.2020 19:50

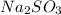 and 9.3 g of
and 9.3 g of 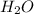 were formed
were formed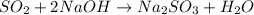
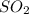 = 64 g
= 64 g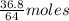 of
of 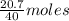 of NaOH = 0.5175 moles of NaOH
of NaOH = 0.5175 moles of NaOH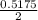 moles of
moles of 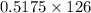 ) g = 65.2 g
) g = 65.2 g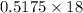 ) g = 9.3 g
) g = 9.3 g

