Chemistry, 22.08.2019 18:10 ggdvj9gggsc
Given the reaction: h2o2(l) ⇌ h2(g) + o2(g) the forward reaction is endothermic. determine which of the following changes would result in equilibrium shifting towards the products. i. increase h2 ii. decrease o2 iii. add a catalyst iv. increase the temperature v. increase h2o2

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 18:00
Acylinder is filled with 2.00 moles of nitrogen, 3.00 moles of argon and 5.00 moles of helium. if the gas mixture is at stp, what is the partial pressure of the argon
Answers: 1

Chemistry, 22.06.2019 01:30
Aroller coaster car is traveling down a track at 22 m/s. the car has a mass of 2000 kg. what is the kinetic energy of the car? a) 22,000 j b) 968,000 j c) 484,000 j d) 44,000 j
Answers: 2

Chemistry, 22.06.2019 02:30
Which element forms an ionic bond with flourine? 1) fluorine 2) carbon 3) potassium 4) oxygen
Answers: 1

Chemistry, 22.06.2019 10:40
Which buffer would be better able to hold a steady ph on the addition of strong acid, buffer 1 or buffer 2? explain. buffer 1: a solution containing 0.10 m nh4cl and 1 m nh3. buffer 2: a solution containing 1 m nh4cl and 0.10 m nh3
Answers: 1
You know the right answer?
Given the reaction: h2o2(l) ⇌ h2(g) + o2(g) the forward reaction is endothermic. determine which of...
Questions

Chemistry, 23.05.2021 18:40

Biology, 23.05.2021 18:40

Mathematics, 23.05.2021 18:40





Mathematics, 23.05.2021 18:40

Mathematics, 23.05.2021 18:40

Mathematics, 23.05.2021 18:40




English, 23.05.2021 18:40

Computers and Technology, 23.05.2021 18:40

History, 23.05.2021 18:40

History, 23.05.2021 18:40


Spanish, 23.05.2021 18:40

Mathematics, 23.05.2021 18:40

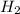 and
and 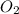 and reactant is
and reactant is