
Chemistry, 22.08.2019 02:20 lisacarter0804
The boiling point of bromine is 59 °c. which of the following best predicts the boiling point of iodine monochloride, a polar compound?
higher than 59 °c because dipole-dipole interactions in iodine monochloride are stronger than dispersion forces in bromine.
lower than 59 °c because ionic bonding in bromine is stronger than covalent bonding in iodine monochloride.
lower than 59 °c because dipole-dipole interactions in iodine monochloride are weaker than in bromine.
higher than 59 °c because ionic bonding in iodine monochloride is stronger than h-bonding in bromine.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 19:00
Describe the chemical reaction based on the chemical equation below. also, explain whether the equation is balanced.
Answers: 1

Chemistry, 22.06.2019 00:00
Which type of bonding involves the complete transfer of a valence electron from a less electrogrative atom to a more electronegative one
Answers: 1

Chemistry, 22.06.2019 00:20
Use the gizmo to find the concentration of the mystery ch3cooh. use the titrant and indicator shown below perform the titration. what is the titrant volume? titrant analyte indicator titrant volume analyte concentration naoh ch3cooh phenophthalein select one: a. 20.0 ml b. 27.0 ml c. 30.0 ml d. 24.0 ml
Answers: 2

Chemistry, 22.06.2019 12:30
What metric units would you use to measure the thickness of a key
Answers: 3
You know the right answer?
The boiling point of bromine is 59 °c. which of the following best predicts the boiling point of iod...
Questions

Mathematics, 28.02.2021 01:00






Mathematics, 28.02.2021 01:00

Mathematics, 28.02.2021 01:00

Mathematics, 28.02.2021 01:00




English, 28.02.2021 01:00

Mathematics, 28.02.2021 01:00



Mathematics, 28.02.2021 01:00

Social Studies, 28.02.2021 01:00


Chemistry, 28.02.2021 01:00

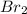 ) is a non-polar molecule due to the absence of a permanent dipole. The only force of attraction in non-polar molecules are the weak dispersion forces.
) is a non-polar molecule due to the absence of a permanent dipole. The only force of attraction in non-polar molecules are the weak dispersion forces. 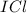 ) is a polar molecule which are held together dipole-dipole forces that are stronger than the London dispersion forces.
) is a polar molecule which are held together dipole-dipole forces that are stronger than the London dispersion forces. 

