
Asolution is 0.010 m in each of pb(no3)2, mn(no3)2, and zn(no3)2. solid naoh is added until the ph of the solution is 8.50. which of the following statements is true? salt ksp pb(oh)2 1.4 × 10–20 mn(oh)2 2.0 × 10–13 zn(oh)2 2.1 × 10–16 a. all three hydroxides will precipitate. b. no precipitate will form. c. only pb(oh)2 will precipitate. d. only mn(oh)2 will precipitate. e. only zn(oh)2 and pb(oh)2 will precipitate.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 02:00
The alkali metals (group 1) consist of lithium (3), sodium (11), potassium (19), rubidium (37), cesium (55), and francium (87). they are soft, metallic solids with low densities and low melting points. based on the data shown in figure 1, how many valence electrons do alkali metals share?
Answers: 3

Chemistry, 22.06.2019 06:10
Explain the relationship between forward and backward reactions in equilibrium, and predict how changing the amount of a reactant (creating a tension) will affect that relationship.
Answers: 1

Chemistry, 22.06.2019 08:30
How would the number of moles (n) of o2 change if the atmospheric pressure doubled but all other variables stayed the same
Answers: 2

You know the right answer?
Asolution is 0.010 m in each of pb(no3)2, mn(no3)2, and zn(no3)2. solid naoh is added until the ph o...
Questions


Mathematics, 24.03.2020 00:25





Mathematics, 24.03.2020 00:25






Mathematics, 24.03.2020 00:25






Computers and Technology, 24.03.2020 00:25

Computers and Technology, 24.03.2020 00:25

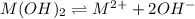
![K_{sp}=[M^{2+}][OH^{-}]^{2}](/tpl/images/0183/5838/c78d3.png)
![[M^{2+}]](/tpl/images/0183/5838/437cc.png) and
and ![[OH^{-}]^{2}](/tpl/images/0183/5838/53c7a.png) exceeds
exceeds 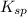 values of given hydroxides then hydroxides of given metal ions will precipitate.
values of given hydroxides then hydroxides of given metal ions will precipitate.![-log[OH^{-}]](/tpl/images/0183/5838/04732.png) = 5.50
= 5.50![[OH^{-}]](/tpl/images/0183/5838/e46dd.png) =
= 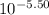
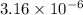
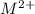 ) are 0.010 M
) are 0.010 M![[M^{2+}][OH^{-}]^{2}=0.010\times (3.16\times 10^{-6})^{2}=3.16\times 10^{-8}](/tpl/images/0183/5838/d1788.png)


