Chemistry, 20.08.2019 23:30 ricardorendon100
Consider the following reaction where kc = 154 at 298 k.2no(g) + br2(g) 2nobr(g)a reaction mixture was found to contain 4.64×10-2 moles of no(g), 4.56×10-2 moles of br2(g) and 0.102 moles of nobr(g), in a 1.00 liter container. is the reaction at equilibrium? if not, what direction must it run in order to reach equilibrium? the reaction quotient, qc, equals .the reactiona. must run in the forward direction to reach equilibrium. b. must run in the reverse direction to reach equilibrium. c. is at equilibrium.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 19:30
Which answer lists the fundamental forces in order from strongest to weakest
Answers: 1

Chemistry, 22.06.2019 03:30
Calculate the molar mass of aluminum oxide (al2o3). express your answer to four significant figures.
Answers: 1

Chemistry, 23.06.2019 13:30
Did you mention that adding weight increased the pressure
Answers: 2

Chemistry, 23.06.2019 13:50
Which compounds are the brønsted-lowry bases in this equilibrium: hc2o4– + h2bo3– h3bo3 + c2o42– ? a. h2bo3– and h3bo3 b. hc2o4– and h3bo3 c. hc2o4– and c2o42– d. h2bo3– and c2o42– e. hc2o4– and h2bo3–
Answers: 2
You know the right answer?
Consider the following reaction where kc = 154 at 298 k.2no(g) + br2(g) 2nobr(g)a reaction mixture w...
Questions


Mathematics, 12.05.2021 22:10





History, 12.05.2021 22:10



Mathematics, 12.05.2021 22:10



History, 12.05.2021 22:10



Chemistry, 12.05.2021 22:10

Mathematics, 12.05.2021 22:10

Mathematics, 12.05.2021 22:10

Physics, 12.05.2021 22:10

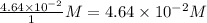
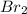 =
= 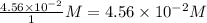
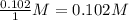
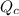 , for this reaction =
, for this reaction = ![\frac{[NOBr]^{2}}{[NO]^{2}[Br_{2}]}](/tpl/images/0183/0904/2c141.png)
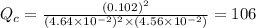
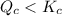 therefore reaction must run in forward direction to increase
therefore reaction must run in forward direction to increase 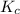 .
.