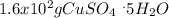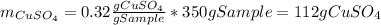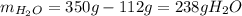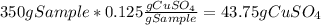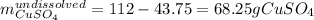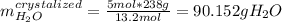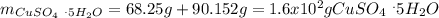Asaturated solution prepared at 70∘c contains 32.0 g cuso4 per 100.0 g solution. a 350 −g sample of this solution is then cooled to 0∘c and cuso4⋅5h2o crystallizes out. if the concentration of a saturated solution at 0∘c is 12.5 gcuso4/100 g soln, what mass of cuso4⋅5h2o would be obtained? [hint: note that the solution composition is stated in terms of cuso4 and that the solid that crystallizes is the hydrate cuso4⋅5h2o.] express your answer using two significant figures.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 08:30
Which part of earth’s surface receives the most direct rays from the sun? a) equator b) ocean c) poles d) mountains
Answers: 2

Chemistry, 22.06.2019 23:00
What is a substance? a. a physical property of matter b. a chemical property of matter c. an element or compound that cannot be physically separated d. characteristics used to tell the difference between mixtures
Answers: 1


Chemistry, 23.06.2019 13:30
How does water evaporating from a glass show that matter is made up of particles? a. the heat energy from the air causes the glass to fill up with water particles. b. the liquid water particles turn into water vapor that spreads in the air. c. the particles of the glass dissolve in water and cause it to evaporate. d. the tiny particles of the glass evaporate and seem to disappear.
Answers: 2
You know the right answer?
Asaturated solution prepared at 70∘c contains 32.0 g cuso4 per 100.0 g solution. a 350 −g sample of...
Questions

Mathematics, 03.04.2020 01:22



Mathematics, 03.04.2020 01:22


History, 03.04.2020 01:22




Social Studies, 03.04.2020 01:22

Biology, 03.04.2020 01:22

Biology, 03.04.2020 01:22

Social Studies, 03.04.2020 01:22



Mathematics, 03.04.2020 01:23

Mathematics, 03.04.2020 01:23

Mathematics, 03.04.2020 01:23

Social Studies, 03.04.2020 01:23