
Chemistry, 20.08.2019 22:20 notsosmart249
Identify whether each species functions as a brønsted-lowry acid or a brønsted-lowry base in this net ionic equation. ch3coo-+hco3-ch3cooh+co32-brønsted- lowrybrønsted-lowrybrønsted-lowrybr ønsted-lowryin this reaction: the formula for the conjugateof ch3coo- isthe formula for the conjugateof hco3- is

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 19:00
Does the temperature affect the solubility of sugar and salt in water? if it does tell me like different temperatures with different solubilities so i can sketch down a graph
Answers: 2



Chemistry, 22.06.2019 07:10
An experimental procedure requires a 10 ml of acid to be dissolved
Answers: 2
You know the right answer?
Identify whether each species functions as a brønsted-lowry acid or a brønsted-lowry base in this ne...
Questions


Biology, 25.09.2019 08:30


Mathematics, 25.09.2019 08:30


Biology, 25.09.2019 08:30


Biology, 25.09.2019 08:30

Mathematics, 25.09.2019 08:30


Mathematics, 25.09.2019 08:30


English, 25.09.2019 08:30


Social Studies, 25.09.2019 08:30




English, 25.09.2019 08:30

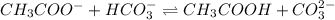
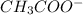 and
and 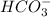 are act as Bronsted Lowry base and acid respectively and
are act as Bronsted Lowry base and acid respectively and 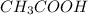 and
and 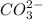 are act as Bronsted Lowry acid and base respectively.
are act as Bronsted Lowry acid and base respectively.

