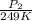The vapor pressure of liquid iodomethane, ch3i, is 40.0 mm hg at 249 k. a sample of ch3i is placed in a closed, evacuated container of constant volume at a temperature of 404 k. it is found that all of the ch3i is in the vapor phase and that the pressure is 72.0 mm hg. if the temperature in the container is reduced to 249 k, which of the following statements are correct
choose all that apply
a. the pressure in the container will be 104 mm hg.
b. no condensation will occur.
c. some of the vapor initially present will condense.
d. only pentane vapor will be present.
e. liquid pentane will be present.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 14:30
The length of a vector arrow represents its magnitude and the point represents its direction true or false apex
Answers: 3

Chemistry, 22.06.2019 07:30
According to the vsepr theory what is the shape of a molecule that has a central atom valence three other items with no lone pairs of electrons
Answers: 1

Chemistry, 22.06.2019 11:40
Enzymes affect the reactions in living cells by changing the
Answers: 3

You know the right answer?
The vapor pressure of liquid iodomethane, ch3i, is 40.0 mm hg at 249 k. a sample of ch3i is placed i...
Questions

Mathematics, 14.12.2020 20:50





Mathematics, 14.12.2020 20:50

Mathematics, 14.12.2020 20:50

Mathematics, 14.12.2020 20:50

Mathematics, 14.12.2020 20:50




Mathematics, 14.12.2020 20:50


Social Studies, 14.12.2020 20:50





Law, 14.12.2020 20:50

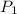 = 72.0 mm Hg,
= 72.0 mm Hg, 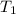 = 404 K
= 404 K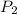 = ? ,
= ? , 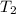 = 249 K
= 249 K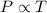 (at constant volume)
(at constant volume)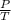 = k
= k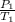 =
= 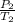
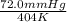 =
=