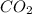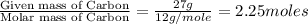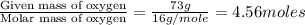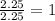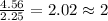Chemistry, 19.08.2019 22:10 lindsey0456
What is the empirical formula of an unknown compound if was determined by a laboratory experiment that it is made up of 27% by mass of carbon and 73% by mass of oxygen?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 10:00
The reactions shown here can be combined to make the overall reaction c(s) + h2o(g) ⇌ co(g) + h2(g) by reversing some and/or dividing all the coefficients by a number. a. c(s) + o2(g) → co2(g) k=1.363×10^69 b. 2 h2(g) + o2(g) → 2 h2o(g) k=1.389×10^80 c. 2co(g) + o2 (g) → 2 co2(g) k=1.477×10^90
Answers: 1

Chemistry, 22.06.2019 15:00
What is the most important factor in determining climates.
Answers: 1

Chemistry, 23.06.2019 01:00
What type of chemical bond is formed between two atoms of bromine 1. metallic 2. hydrogen 3. ionic 4. covalent
Answers: 1

You know the right answer?
What is the empirical formula of an unknown compound if was determined by a laboratory experiment th...
Questions


Mathematics, 29.02.2020 00:57


















Mathematics, 29.02.2020 00:59