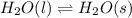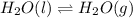Question 2: phase changes (14 points) a. rewrite each of the following equations for phase changes, to include the heat required for the phase change. use values from latent heat and specific heat constant tables when necessary. indicate whether each phase change is endothermic or exothermic. (2 points) i. h2o(l) h2o(s) (1 point) ii. h2o(l) h2o(g) (1 point)

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 10:30
Apiece of metal with a length of 1.42 cm was measured using four different instruments. which of the following measurements is the most accurate?
Answers: 3


Chemistry, 22.06.2019 20:00
Suppose that some of the compound spilled out of the crucible after it was heated. would that cause the percent by mass of water in the compound determined by the experiment to be too low, too high, or unchanged? briefly explain your answer.
Answers: 1

Chemistry, 22.06.2019 21:30
If you burn 46.6 g of hydrogen and produce 416 g of water, how much oxygen reacted
Answers: 3
You know the right answer?
Question 2: phase changes (14 points) a. rewrite each of the following equations for phase changes,...
Questions


Mathematics, 19.06.2020 00:57


English, 19.06.2020 00:57





Mathematics, 19.06.2020 01:57


Computers and Technology, 19.06.2020 01:57


Mathematics, 19.06.2020 01:57



Mathematics, 19.06.2020 01:57

Mathematics, 19.06.2020 01:57

Chemistry, 19.06.2020 01:57

Mathematics, 19.06.2020 01:57