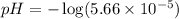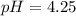Chemistry, 13.08.2019 04:30 jenifferplowman
The acid dissociation ka of propionic acid c2h5co2h is 1.310x10−5. calculate the ph of a 3.010x10−4m aqueous solution of propionic acid. round your answer to 2 decimal places.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 02:00
If you add 10ml of hot water to 10ml of cold water and the change in tempature 8°c calculate how much energy is gained by the cold water
Answers: 1

Chemistry, 22.06.2019 07:30
The scheme below is from a series of reactions that are part of a synthesis of vitamin a. answer the following questions with reference to this scheme. (i) what is "reagent a"? (ii) draw a step-by-step mechanism which explains the formation of compound c from compound b (iii) which reagents would you use to form compound e from compounds c and d (reagents b and c)? for each reagent suggested above in (ii) explain the role of the reagent in the reaction to (iv) form compound e. you may wish to do this by drawing a mechanism. 1. addition of reagent a но reagent a 2. н,о" thо oh нон-с compound a. compound b. compound c .ch-оh 1. reagent b "сно 2. reagent c сh oh compound e. compound d.
Answers: 2

Chemistry, 22.06.2019 13:10
What type of interaction occurs between the r groups of valine and isoleucine in a tertiary structure? view available hint(s) what type of interaction occurs between the r groups of valine and isoleucine in a tertiary structure? salt bridge disulfide bridge hydrogen bond hydrophobic interaction
Answers: 1

You know the right answer?
The acid dissociation ka of propionic acid c2h5co2h is 1.310x10−5. calculate the ph of a 3.010x10−4m...
Questions




English, 19.10.2020 08:01

Mathematics, 19.10.2020 08:01




Mathematics, 19.10.2020 08:01

Biology, 19.10.2020 08:01




Social Studies, 19.10.2020 08:01




History, 19.10.2020 08:01

English, 19.10.2020 08:01

Mathematics, 19.10.2020 08:01

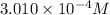
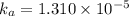
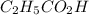 (weak acid) is,
(weak acid) is,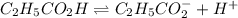
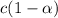
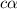
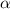 is degree of dissociation
is degree of dissociation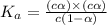
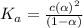
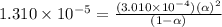
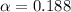
![[H^+]=c\alpha=(3.010\times 10^{-4})\times (0.188)=5.66\times 10^{-5}M](/tpl/images/0174/7681/497f7.png)
![pH=-\log [H^+]](/tpl/images/0174/7681/37e81.png)