
Apossible mechanism for the gas phase reaction of no and h2 is as follows: step 1 2no n2o2 step 2 n2o2 + h2 n2o + h2o step 3 n2o + h2 n2 + h2o which of the following statements concerning this mechanism is not directly supported by the information provided? a. n2o2 is an intermediate. b. all steps are bimolecular reactions. c. the rate expression for step 1 is rate = k[no]2. d. step 1 is the rate determining step. e. there is no catalyst in this reaction. 1 points question 9

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 01:00
Water is important for the of cells. a: size, shape, and temperature b: temperature, color, and odor c: color, odor, and size d: shape, temperature, and color
Answers: 2

Chemistry, 22.06.2019 11:00
Ais a mountain created from eruptions of lava, ash, rocks, and hot gases.
Answers: 1

Chemistry, 22.06.2019 13:30
Some animals that try to adapt to climate changes eventually die due to starvation, as climate change alters the web.
Answers: 2

Chemistry, 22.06.2019 14:30
Consider the reduction reactions and their equilibrium constants. cu+(aq)+e−↽−−⇀cu(s)pb2+(aq)+2e−↽−−⇀pb(s)fe3+(aq)+3e−↽−−⇀fe(=6.2×108=4.0×10−5=9.3×10−3 cu + ( aq ) + e − ↽ − − ⇀ cu ( s ) k =6.2× 10 8 pb 2 + ( aq ) +2 e − ↽ − − ⇀ pb ( s ) k =4.0× 10 − 5 fe 3 + ( aq ) +3 e − ↽ − − ⇀ fe ( s ) k =9.3× 10 − 3 arrange these ions from strongest to weakest oxidizing agent.
Answers: 3
You know the right answer?
Apossible mechanism for the gas phase reaction of no and h2 is as follows: step 1 2no n2o2 step 2 n...
Questions


English, 04.07.2019 00:30

English, 04.07.2019 00:30

Social Studies, 04.07.2019 00:30


English, 04.07.2019 00:30


Biology, 04.07.2019 00:30








Computers and Technology, 04.07.2019 00:30


Mathematics, 04.07.2019 00:30

Mathematics, 04.07.2019 00:30

Mathematics, 04.07.2019 00:30

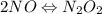 (fast)
(fast)![[NO]^{2}](/tpl/images/0174/7336/f0d3d.png)

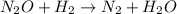 (fast)
(fast) is intermediate in nature.
is intermediate in nature.

