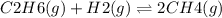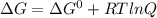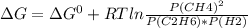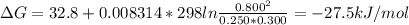For a gaseous reaction, standard conditions are 298 k and a partial pressure of 1 atm for all species. for the reaction c2h6(g)+h2(g)↽−−⇀2ch4(g) the standard change in gibbs free energy is δ°=−32.8 kj/mol . what is δg for this reaction at 298 k when the partial pressures are =0.250 atm , =0.300 atm , and =0.800 atm ?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 03:50
Which of the following statements about acidic water is true? a. acid has no effect on the h,o molecules. b. the solution contains a larger number of oh ions than h,o ions. c. the solution contains a larger number of h,o ions than qh ions. d. the solution contains an equal number of h,o ions and oh ions. none of the above e.
Answers: 1


Chemistry, 22.06.2019 14:10
13. a covalent bond between two atoms is likely to be polar if: a. one of the atoms is much more electronegative than the other. b. the two atoms are equally electronegative. c. the two atoms are of the same element. d. the bond is part of a tetrahedrally shaped molecule. e. one atom is an anion.
Answers: 1

Chemistry, 23.06.2019 06:40
8. how much enthalpy/heat is transferred when 0.5113gof ammonia (nh3) reacts with excess oxygen according| to the following equation: 4nh3 +502 - 4n0+ 6h20ah = -905.4j
Answers: 1
You know the right answer?
For a gaseous reaction, standard conditions are 298 k and a partial pressure of 1 atm for all specie...
Questions

Social Studies, 21.07.2020 08:01




Mathematics, 21.07.2020 08:01

Law, 21.07.2020 08:01

Computers and Technology, 21.07.2020 08:01






History, 21.07.2020 08:01

English, 21.07.2020 08:01

Biology, 21.07.2020 08:01

Mathematics, 21.07.2020 08:01

Mathematics, 21.07.2020 08:01